
A popular anti-aging therapy produced dramatic brain changes in mice.
A drug pairing often promoted in anti-aging research may come with an unexpected cost in the brain. University of Connecticut scientists report in PNAS that dasatinib+quercetin (D+Q) caused significant brain damage in mice, including the loss of myelin, the fatty covering that helps nerve cells send signals quickly and efficiently.
The finding raises an important warning for doctors considering the treatment as a preventive therapy, but it also offers a potentially useful new clue for understanding multiple sclerosis.
“When you administer this cocktail to an animal, young or old, the myelin is damaged, which makes it disappear. Even worse in the young animals” than in the aged ones, says UConn School of Medicine immunologist Stephen Crocker.
Myelin is the protective coating around nerves. Without it, nerves cannot send signals as effectively. That can lead to numbness, pain, trouble walking, and problems with memory and thinking. Loss of myelin is the main driver of multiple sclerosis. Crocker and his team found the same kind of damage in mice treated with dasatinib+quercetin (D+Q), using doses commonly studied for aging-related inflammation and metabolic disorders.
A Popular Treatment With Limited Brain Research
D+Q has become a widely studied drug combination in anti-aging research. Many studies suggest it can help remove aged cells that contribute to inflammation and other age-related problems. Researchers are testing it for conditions that include type II diabetes and Alzheimer’s. Some people interested in anti-aging also take it off-label, even though medical experts advise against that use.
Still, very little research has examined how it affects the brain.
Evan Lombardo ’23 (CLAS), now a Dartmouth neuroscience graduate student, and Robert Pijewski ’21 Ph.D., now at Anna Maria College, were working in Crocker’s lab when they asked whether D+Q might help rejuvenate the brains of people with multiple sclerosis and possibly ease their symptoms.
To test that idea, they gave the treatment to both young (6 to 9 months) and old (22 months) mice. They also studied brain cells grown in the lab. Those cells were oligodendrocytes, which are responsible for producing and maintaining myelin.
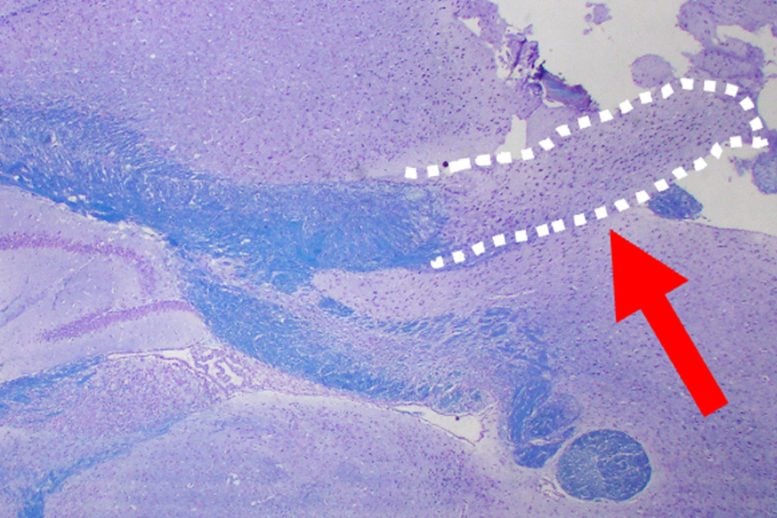
Dramatic Changes in Myelin and Brain Structure
The results were clear. In healthy mice, myelin surrounds axons (nerve cells) in the brain and appears as dark rings around the lighter axon. After treatment with D+Q, the mice had far less myelin around their axons. The damage was more severe in the younger mice.
The corpus callosum, which links the cerebral cortex with other parts of the brain and supports many important functions, also disappeared in mice treated with D+Q. Similar damage is sometimes seen in people who have received chemotherapy, where it is linked to symptoms often called “chemo brain.”
When the researchers closely examined the injured brain tissue, they found clues about what had happened. The oligodendrocytes had not died. Instead, they had shifted back into a more juvenile state. The cells also showed abnormal metabolism.
“We suspect the drugs are choking off energy the cells need, and the cells respond by reducing complexity, reverting to a younger state, but less functional,” Crocker says.
New Clues About Multiple Sclerosis
These reverted cells closely resemble a distinct group of cells found in people with multiple sclerosis. That suggests myelinating cells in multiple sclerosis may come under stress and fall back to an earlier stage of development. It also raises the possibility that those cells could recover. That is what the researchers are now trying to find out.
“If we can mimic this, we have an amazing opportunity to see if the cells can recover and repair the brain,” Crocker says.
Reference: “Senolytic treatment induces oligodendrocyte dysfunction and demyelination in the corpus callosum” by Evan R. Lombardo, Robert S. Pijewski, Jake T. Lustig, Zaenab Dhari, Anirudhya Lahiri, Lucille E. Papile, Erica R. Lavoie, Vanessa M. Scanlon, Jenna M. Bartley and Stephen J. Crocker, 17 March 2026, Proceedings of the National Academy of Sciences.
DOI: 10.1073/pnas.2524897123
Never miss a breakthrough: Join the SciTechDaily newsletter.
Follow us on Google and Google News.
2 Comments
This must be true since the effect has happened with mice. We are in a rat race and even if we win, we still get to be a rat. So be careful because this might apply to us. Pásame queso, por favor. ¡Échele Primero!
Knowledge graph analysis of dasatinib and quercetin’s molecular targets using the INDRA CoGEx biomedical knowledge graph reveals at least five convergent mechanisms that explain the demyelination reported by Lombardo, Pijewski, and colleagues. Dasatinib inhibits 84 kinases, several of which are critical to oligodendrocyte biology: FYN (a Src-family kinase in the Reactome “Nervous system development” pathway required for OPC-to-oligodendrocyte maturation), PDGFRα (the canonical OPC survival receptor, expressed in the CNS with associated neurodevelopmental delay phenotypes), and CSF1R — whose loss-of-function mutations cause Hereditary Diffuse Leukoencephalopathy with Spheroids, a white matter disease characterized by cerebral white matter atrophy, thin corpus callosum, and CNS axonal spheroids, closely mirroring the pathology reported here. Dasatinib also inhibits ErbB3/4, disrupting neuregulin-mediated myelination signaling, and all four p38 MAPK isoforms, potentially blocking the stress-adaptive responses oligodendrocytes need under metabolic challenge. Quercetin compounds the damage by inhibiting casein kinase 2 (CK2), which participates in both phosphatidylcholine synthesis (a major myelin membrane lipid) and Wnt signaling that governs OPC differentiation timing. Critically, all five target kinases — FYN, PDGFRα, CSF1R, ErbB3, and ErbB4 — show confirmed expression across cortical regions and spinal cord in the INDRA tissue atlas. Together, these drugs simultaneously block oligodendrocyte maturation signals (FYN, ErbB), survival cues (PDGFRα), microglial trophic support (CSF1R), and the metabolic machinery for myelin lipid biosynthesis (CK2/PI3K/AKT), creating conditions under which de-differentiation to a juvenile state — rather than apoptosis — is the only viable cellular response, consistent with the authors’ observations and with dasatinib’s existing clinical side-effect profile, which already includes neuropathy, optic neuritis, amnesia, and confusion.