Scientists set a record for the highest conversion rate of carbon dioxide at low temperatures with copper-modified indium oxide, signifying sustainable e-fuel.
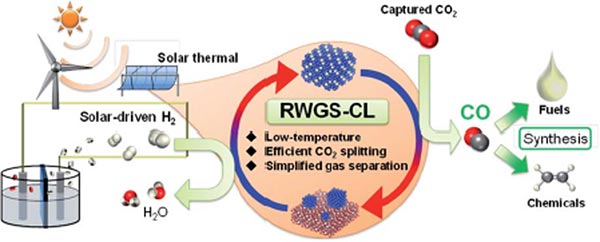
With ever-worsening climate change, there is a growing need for technologies that can capture and use up the atmospheric CO2 (carbon dioxide) and reduce our carbon footprint. Within the realm of renewable energy, CO2-based e-fuels have emerged as a promising technology that attempts to convert atmospheric CO2 into clean fuels. The process involves production of synthetic gas or syngas (a mixture of hydrogen and carbon monoxide (CO)). With the help of the reverse water-gas shift (RWGS) reaction, CO2 is broken down into the CO necessary for syngas. While promising in its conversion efficiency, the RWGS reaction requires incredibly high temperatures (>700°C or 1300°F) to proceed, while also generating unwanted byproducts.
To tackle these problems, scientists developed a modified chemical-looping version of the RWGS reaction that converts CO2 to CO in a two-step method. First, a metal oxide, used as an oxygen storage material, is reduced by hydrogen. Subsequently, it is re-oxidized by CO2, yielding CO. This method is free of undesirable byproducts, makes gas separation simpler, and can be made feasible at lower temperatures depending on the oxide chosen. Consequently, scientists have been looking for oxide materials that exhibit high oxidation-reduction rates without requiring high temperatures.
A Breakthrough with Copper-Indium Oxide
In a recent study published in Chemical Science, scientists from Waseda University and ENEOS Corporation in Japan have revealed that a novel indium oxide modified with copper (Cu-In2O3) exhibits a record-breaking CO2 conversion rate of 10 mmolh-1g-1 at relatively modest temperatures (400–500°C or 750-930°F), making it a frontrunner among oxygen storage materials required for low-temperature CO2 conversion. To better understand this behavior, the team investigated the structural properties of Cu-In oxide along with the kinetics involved in the chemical-looping RWGS reaction.
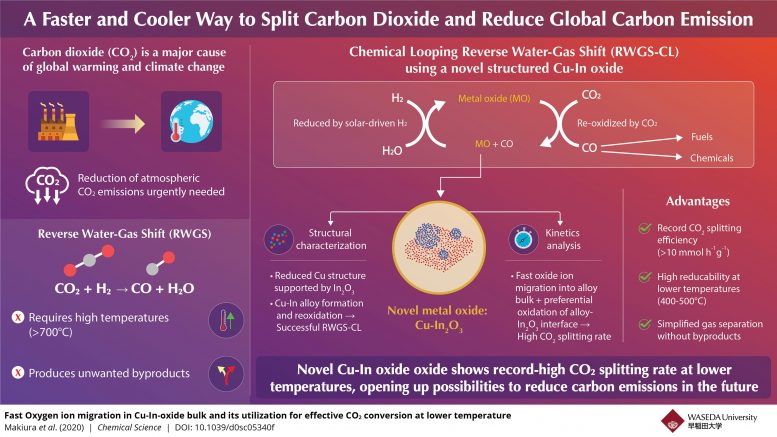
The scientists carried out X-ray-based analyses and found that the sample initially contained a parent material, Cu2In2O5, which was first reduced by hydrogen to form a Cu-In alloy and indium oxide (In2O3) and then oxidized by CO2 to yield Cu-In2O3 and CO. X-ray data further revealed that it underwent oxidation and reduction during the reaction, providing the key clue to scientists. “The X-ray measurements made it clear that the chemically looped RWGS reaction is based on the reduction and oxidation of Indium which leads to the formation and oxidation of the Cu-In alloy,” explains Professor Yasushi Sekine of Waseda University, who led the study.
Fast Oxidation Enables Record CO₂ Conversion Rates
The kinetics investigations provided further insights into the reaction. The reduction step revealed that Cu was responsible for the reduction of indium oxide at low temperatures, while the oxidation step showed that the Cu-In alloy surface preserved a highly reduced state while its bulk got oxidized. This allowed the oxidation to happen twice as quickly as that of other oxides. The team attributed this peculiar oxidation behavior to a rapid migration of negatively charged oxygen ions from the Cu-In alloy surface to its bulk, which assisted in the preferential bulk oxidation.
The results have, quite expectedly, excited scientists about the future prospects of copper-indium oxides. “Given the current situation with carbon emission and global warming, a high-performance carbon dioxide conversion process is greatly desired. Although the chemically looped RWGS reaction works well with many oxide materials, our novel Cu-In-oxide here shows a remarkably higher performance than any of them. We hope that this will contribute significantly to reducing our carbon footprint and driving humankind towards a more sustainable future,” concludes Sekine.
Reference: “Fast oxygen ion migration in Cu–In–oxide bulk and its utilization for effective CO2 conversion at lower temperature” by Jun-Ichiro Makiura, Takuma Higo, Yutaro Kurosawa, Kota Murakami, Shuhei Ogo, Hideaki Tsuneki, Yasushi Hashimoto, Yasushi Satob and Yasushi Sekine, 23 December 2020, Chemical Science.
DOI: 10.1039/d0sc05340f
Never miss a breakthrough: Join the SciTechDaily newsletter.
Follow us on Google and Google News.
1 Comment
These kinds of studies look at technological possibilities without considering the big picture. While copper is relatively abundant, modern deposits are very low grade and require huge amounts of energy to extract and process. This inevitably produces a large amount of CO2! Indium is not as abundant, but still requires a lot of energy to obtain. One has to be careful that the ‘solution’ to a perceived problem doesn’t become a treadmill where we are just running in place.