The new drug circumvents the neurotoxic inflammatory side effects that occur with conventional treatments.
Aduhelm, a monoclonal antibody that targets amyloid beta (A), recently became the first United States Food and Drug Administration (FDA)-approved drug for Alzheimer’s disease (AD) based on its ability to decrease Aβ plaque burden in AD patients. However, its impact on cognitive improvement is still disputed.
Furthermore, approximately 40% of patients treated with this antibody experienced serious side effects such as cerebral edemas (ARIA-E) and hemorrhages (ARIA-H), which are likely related to inflammatory responses in the brain caused by the Aβ antibody binding Fc receptors (FCR) of immune cells such as microglia and macrophages.
These inflammatory side effects have the potential to worsen cognitive impairment in Alzheimer’s disease patients by causing neuronal cell death and synapse elimination by activated microglia. As a result of its inflammatory side effects, current Aβ antibody-based immunotherapy has the risk of inflicting more damage than good.
Aβ-Gas6: A New Fusion Protein for Aβ Clearance
To address these issues, a team of researchers at the Korea Advanced Institute of Science and Technology (KAIST) in South Korea created Aβ-Gas6, a new fusion protein drug that successfully eliminates Aβ through a completely different mechanism than Aβ antibody-based immunotherapy. In a mouse model of Alzheimer’s disease, A-Gas6 not only eliminated Aβ more effectively, but it also avoided the neurotoxic inflammatory side effects associated with conventional antibody treatments.
The findings were recently published in the journal Nature Medicine.
Utilizing Efferocytosis to Avoid Inflammatory Responses
“FcR activation by Aβ targeting antibodies induces microglia-mediated Aβ phagocytosis, but it also produces inflammatory signals, inevitably damaging brain tissues,” said paper authors Chan Hyuk Kim and Won-Suk Chung, associate professors in the Department of Biological Sciences at KAIST.
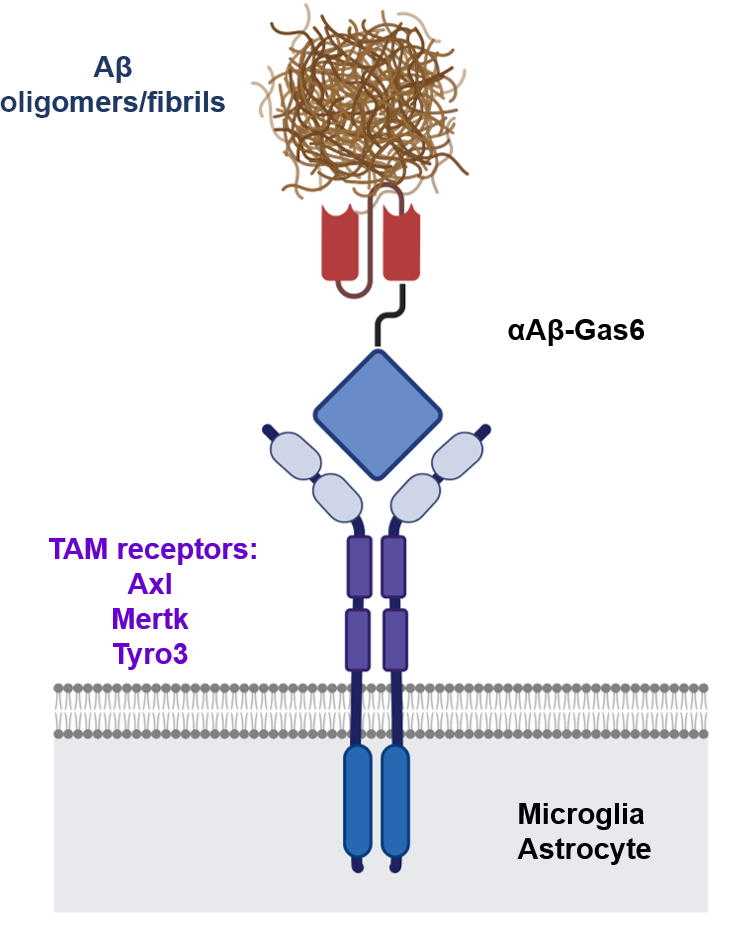
“Therefore, we utilized efferocytosis, a cellular process by which dead cells are removed by phagocytes as an alternative pathway for the clearance of Aβ in the brain,” Prof. Kim and Chung said. “Efferocytosis is accompanied by anti-inflammatory responses to maintain tissue homeostasis. To exploit this process, we engineered Gas6, a soluble adaptor protein that mediates efferocytosis via TAM phagocytic receptors in such a way that its target specificity was redirected from dead cells to Aβ plaques.”
Effectiveness of Aβ-Gas6 in Alzheimer’s Disease Models
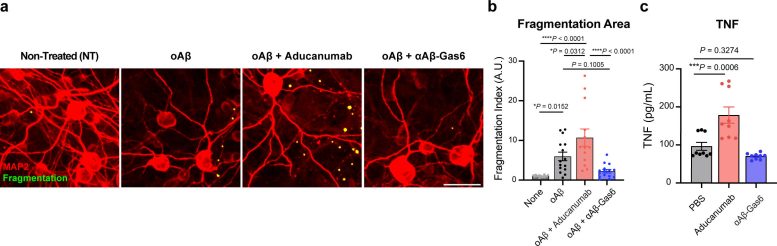
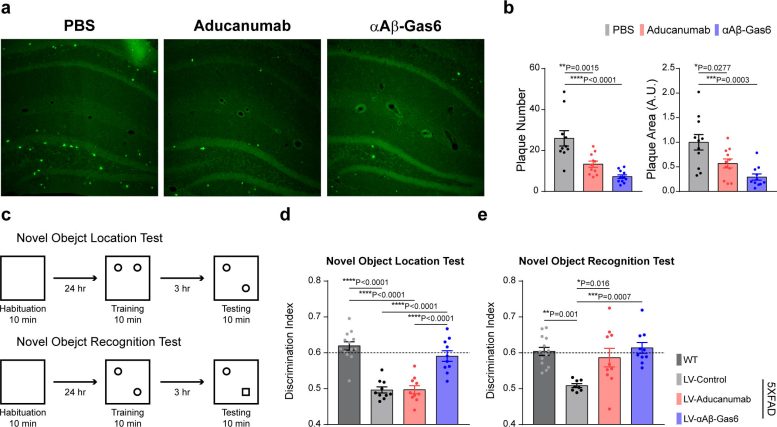
The professors and their team demonstrated that the resulting αAβ-Gas6 induced Aβ engulfment by activating not only microglial but also astrocytic phagocytosis since TAM phagocytic receptors are highly expressed by these two major phagocytes in the brain. Importantly, αAβ-Gas6 promoted the robust uptake of Aβ without showing any signs of inflammation and neurotoxicity, which contrasts sharply with the treatment using an Aβ monoclonal antibody. Moreover, they showed that αAβ-Gas6 substantially reduced excessive synapse elimination by microglia, consequently leading to better behavioral rescues in AD model mice.
“By using a mouse model of cerebral amyloid angiopathy (CAA), a cerebrovascular disorder caused by the deposition of Aβ within the walls of the brain’s blood vessels, we also showed that the intrathecal administration of Gas6 fusion protein significantly eliminated cerebrovascular amyloids, along with a reduction of microhemorrhages. These data demonstrate that aAb-Gas6 is a potent therapeutic agent in eliminating Aβ without exacerbating CAA-related microhemorrhages.”
Professors Kim and Chung noted, “We believe our approach can be a breakthrough in treating AD without causing inflammatory side effects and synapse loss. Our approach holds promise as a novel therapeutic platform that is applicable to more than AD. By modifying the target-specificity of the fusion protein, the Gas6-fusion protein can be applied to various neurological disorders as well as autoimmune diseases affected by toxic molecules that should be removed without causing inflammatory responses.”
Reference: “Anti-inflammatory clearance of amyloid-β by a chimeric Gas6 fusion protein” by Hyuncheol Jung, Se Young Lee, Seongjoon Lim, Hyeong Ryeol Choi, Yeseong Choi, Minjin Kim, Segi Kim, Yujean Lee, Kyung Ho Han, Won-Suk Chung and Chan Hyuk Kim, 4 August 2022, Nature Medicine.
DOI: 10.1038/s41591-022-01926-9
Professors Kim and Chung founded “Illimis Therapeutics” based on this strategy of designing chimeric Gas6 fusion proteins that would remove toxic aggregates from the nervous system. Through this company, they are planning to further develop various Gas6-fusion proteins not only for Ab but also for Tau to treat AD symptoms.
This study was funded by the Korea Health Industry Development Institute, the Korea Dementia Research Center, the Korean Ministry of Health and Welfare, and the Korean Ministry of Science and ICT.
Never miss a breakthrough: Join the SciTechDaily newsletter.
Follow us on Google and Google News.