
A team of scientists led by James Godwin, Ph.D., of the MDI Biological Laboratory in Bar Harbor, Maine, has come a step closer to unraveling the mystery of why salamanders can regenerate while adult mammals cannot with the discovery of differences in molecular signaling that promote regeneration in the axolotl, a highly regenerative salamander, while blocking it in the adult mouse. Godwin is pictured here with a tank containing an axolotl. Credit: MDI Biological Laboratory
Discovery in Salamanders by James W. Godwin, Ph.D., brings science closer to the development of regenerative medicine therapies.
Many salamanders can readily regenerate a lost limb, but adult mammals, including humans, cannot. Why this is the case is a scientific mystery that has fascinated observers of the natural world for thousands of years.
Now, a team of scientists led by James Godwin, Ph.D., of the MDI Biological Laboratory in Bar Harbor, Maine, has come a step closer to unraveling that mystery with the discovery of differences in molecular signaling that promote regeneration in the axolotl, a highly regenerative salamander, while blocking it in the adult mouse, which is a mammal with limited regenerative ability.
“Scientists at the MDI Biological Laboratory have been relying on comparative biology to gain insights into human health since its founding in 1898,” said Hermann Haller, M.D., the institution’s president. “The discoveries enabled by James Godwin’s comparative studies in the axolotl and mouse are proof that the idea of learning from nature is as valid today as it was more than one hundred and twenty years ago.”

The axolotl, a Mexican salamander that is now all but extinct in the wild, is a favorite model in regenerative medicine research because of its one-of-a-kind status as nature’s champion of regeneration. While most salamanders have some regenerative capacity, the axolotl can regenerate almost any body part, including brain, heart, jaws, limbs, lungs, ovaries, spinal cord, skin, tail, and more. A team of scientists led by James Godwin, Ph.D., of the MDI Biological Laboratory has come a step closer to unraveling the mystery of why salamanders can regenerate while adult mammals cannot with the discovery of differences in molecular signaling that promote regeneration in the axolotl, a highly regenerative salamander, while blocking it in the adult mouse. Credit: MDI Biological Laboratory
Instead of regenerating lost or injured body parts, mammals typically form a scar at the site of an injury. Because the scar creates a physical barrier to regeneration, research in regenerative medicine at the MDI Biological Laboratory has focused on understanding why the axolotl doesn’t form a scar — or, why it doesn’t respond to injury in the same way that the mouse and other mammals do.
“Our research shows that humans have untapped potential for regeneration,” Godwin said. “If we can solve the problem of scar formation, we may be able to unlock our latent regenerative potential. Axolotls don’t scar, which is what allows regeneration to take place. But once a scar has formed, it’s game over in terms of regeneration. If we could prevent scarring in humans, we could enhance quality of life for so many people.”
The axolotl as a model for regeneration
The axolotl, a Mexican salamander that is now all but extinct in the wild, is a favorite model in regenerative medicine research because of its one-of-a-kind status as nature’s champion of regeneration. While most salamanders have some regenerative capacity, the axolotl can regenerate almost any body part, including brain, heart, jaws, limbs, lungs, ovaries, spinal cord, skin, tail, and more.
Since mammalian embryos and juveniles have the ability to regenerate — for instance, human infants can regenerate heart tissue and children can regenerate fingertips — it’s likely that adult mammals retain the genetic code for regeneration, raising the prospect that pharmaceutical therapies could be developed to encourage humans to regenerate tissues and organs lost to disease or injury instead of forming a scar.
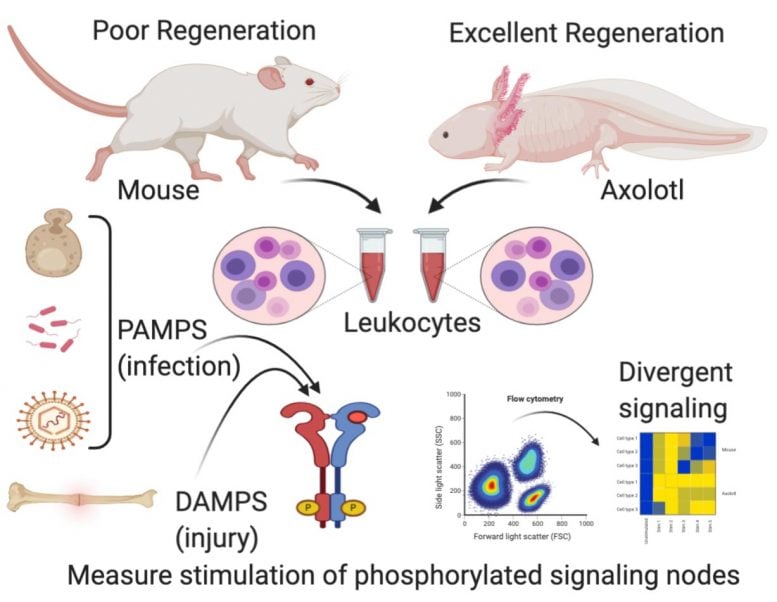
This graphical abstract from James Godwin, Ph.D., of the MDI Biological Laboratory in Bar Harbor, Maine, depicts the divergence between molecular signaling in the immune systems of the axolotl, a Mexican salamander that can readily regenerate limbs and other body parts, and the adult mouse, which cannot. Credit: MDI Biological Laboratory
In his recent research, Godwin compared immune cells called macrophages in the axolotl to those in the mouse with the goal of identifying the quality in axolotl macrophages that promotes regeneration. The research builds on earlier studies in which Godwin found that macrophages are critical to regeneration: when they are depleted, the axolotl forms a scar instead of regenerating, just like mammals.
The recent research found that although macrophage signaling in the axolotl and in the mouse were similar when the organisms were exposed to pathogens such as bacteria, funguses and viruses, when it came to exposure to injury it was a different story: the macrophage signaling in the axolotl promoted the growth of new tissue while that in the mouse promoted scarring.
The paper on the research, entitled “Distinct TLR Signaling in the Salamander Response to Tissue Damage” was recently published in the journal Developmental Dynamics. In addition to Godwin, authors include Nadia Rosenthal, Ph.D., of The Jackson Laboratory; Ryan Dubuque and Katya E. Chan of the Australian Regenerative Medicine Institute (ARMI); and Sergej Nowoshilow, Ph.D., of the Research Institute of Molecular Pathology in Vienna, Austria.

Special Issue: Salamander Models for Elucidating Mechanisms of Developmental Biology, Evolution & Regeneration/Repair: Part One
Godwin, who holds a joint appointment with The Jackson Laboratory, was formerly associated with ARMI and Rosenthal is ARMI’s founding director. The MDI Biological Laboratory and ARMI have a partnership agreement to promote research and education on regeneration and the development of new therapies to improve human health.
Specifically, the paper reported that the signaling response of a class of proteins called toll-like receptors (TLRs), which allow macrophages to recognize a threat such an infection or a tissue injury and induce a pro-inflammatory response, were “unexpectedly divergent” in response to injury in the axolotl and the mouse. The finding offers an intriguing window into the mechanisms governing regeneration in the axolotl.
Being able to ‘pull the levers of regeneration’
The discovery of an alternative signaling pathway that is compatible with regeneration could ultimately lead to regenerative medicine therapies for humans. Though regrowing a human limb may not be realistic in the short term, significant opportunities exist for therapies that improve clinical outcomes in diseases in which scarring plays a major role in the pathology, including heart, kidney, liver, and lung disease.
“We are getting closer to understanding how axolotl macrophages are primed for regeneration, which will bring us closer to being able to pull the levers of regeneration in humans,” Godwin said. “For instance, I envision being able to use a topical hydrogel at the site of a wound that is laced with a modulator that changes the behavior of human macrophages to be more like those of the axolotl.”
Godwin, who is an immunologist, chose to examine the function of the immune system in regeneration because of its role in preparing the wound for repairs as the equivalent of a first responder at the site of an injury. His recent research opens the door to further mapping of critical nodes in TLR signaling pathways that regulate the unique immune environment enabling axolotl regeneration and scar-free repair.
Reference: “Distinct toll-like receptor signaling in the salamander response to tissue damage” by Ryan J. Debuque, Sergej Nowoshilow, Katya E. Chan, Nadia A. Rosenthal and James W. Godwin, 1 April 2021, Developmental Dynamics.
DOI: 10.1002/dvdy.340
Godwin’s research is supported by an Institutional Development Award (IDeA) from the National Institute of General Medical Sciences of the National Institutes of Health under grant numbers P20GM103423 and P20GM104318 to the MDI Biological Laboratory. ARMI is supported by grants from the State Government of Victoria, Australia. The mouse studies were supported by Jackson Laboratory institutional funds.







Let us consider that mammals can regenerate according to Sir Godwin if the scar doesn’t become on the place of injury, but it baffling that if the scar doesn’t form then how will it be possible to protect inner body from germs present in the outer environment 🤔?and also if we want to regenerate the lost part in mammals it may take sometime which is not mentioned here in this post or is unknown.Also unfortunately, if it takes a long time to regenerate the lost body part or organ it is not possible to do the sterilization of the area around lost body part or organ with the help of chemotheraputic agents or antibiotics for such a long time.By the way Dr.Godwin is doing a great job and it will prove beneficial to humans to a great extent.May God help him!
And also different parts of the bodies of mammals will take different times to regenerate themselves,if this happens and all parts or organs of the bodies of mammals take different times to regenerate then the organs with a long regeneration time can’t regenerate because of the limitation that we can’t protect the inner body environment from the outer body environment or inner organ environment from the organ’s outer environment without a barrier for such a long time,so if regeneration is possible then only body parts of mammals with small regeneration time will regenerate themselves.So,if we want all body parts to regenerate themselves then we also have to work on the techniques of the sterilization and make them strong. Also there is another idea that if regeneration of the lost part starts and it produces end,tip,boundary of the lost part first than the part in the middle only then it is possible for the lost body part to regenerate itself to its original shape.To understand it let us have a look on the finger which is cut accidentally, if we want it to regenerate itself safely and to regenerate itself to its old original shape then it should be start regenerating from the nail and it should grow or regenerate itself up just like a plant or tree,if it regenerates the third digit from the top or nail then it will not possible for it to regenerate itself as it was before injury,in the same way if an organ gets cut then it should regenerate its boundary first then it should expand to its original shape,otherwise regeneration may have adverse effects on human bodies instead of benefiting us i.e it will change the shapes of organs and body parts.Randome regeneration or regeneration starting from any place of lost part can only give results if the shapes of body parts or organs are conserved in the genes or DNA.We have to reveal the regeneration potential of DNA and make changes in it for every specific part to regenerate itself.According to me there will be different change in DNA for finger to regenerate itself and separate change in DNA for the eye to regenerate it self. Thanks!…Dr.Godwin keep it up.You are an experienced person and you can do it and you know about this issue better than me.I just gave my point of view about regeneration.I may be proved wrong!I wish to work with you Dr.Godwin.
The ligaments, like the acl or medial or lateral wah originate from the marrow, with the tendons muscular tangents from one to the other like wrappings of fine linen, around the super soft cartilage at the beginning of the skeletal formation, then the connections… and the flexibility.. all associated with the marrow and the structure of the marrow with the ecistestential environment. The relationship between dead weight and the lightness of being. Nice.
In the nearer term such research could lead to cures for fibrotic diseases — from the deadly idiopathic pulmonary fibrosis to the disfiguring Dupuytren’s contracture and Peyronie’s