Until now, cancer researchers haven’t been able to track what errant cancer cells have been doing in patients’ bodies, ie forming tumors. Three different groups studying tumors in mice have accomplished this. Their results support the idea that a small subset of cells drives tumor growth and that curing cancer would require eliminating these rogue cells.
The researchers published their findings in the journals Nature and Science. For now, it’s too soon to tell if the results, obtained for tumors of the brain, gut, and skin, will apply to other cancers, states Luis Parada, a cancer researcher at the University of Texas Southwestern Medical Center in Dallas, Texas, and who led the study on the brain tumors. If they do, there will be a paradigm shift in the way that chemotherapy efficacy is evaluated and how new therapeutics are developed, he states.
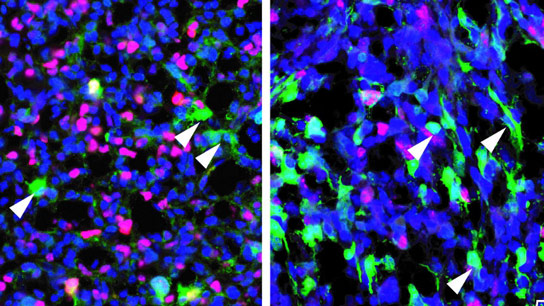
What underlies this scenario is that many tumors might be fueled by cancer stem cells, which produce other types of cancer cells, just as ordinary stem cells produce normal tissue. All three groups tried to address a knowledge gap using genetic techniques to track cancer cells. Parada and his team began by testing whether a genetic marker that labels healthy adult neural stem cells but not their specialized descendants might also label cancer stem cells in glioblastoma. They discovered that all tumors contain at least a few labeled cells, presumably the cancer’s stem cells. Tumors also contained many unlabeled cells, which could be killed off with standard chemotherapy, but the tumors quickly returned. The unlabeled cells originated from their labeled predecessors. Once chemotherapy was paired with a genetic therapy to suppress the labeled cells, the tumors shrank back into residual vestiges.

Hans Clevers, a stem-cell biologist at the Hubrecht Institute in Utrecht, the Netherlands, focused on gut-based cancers. Previously, Clevers and his team had shown that a genetic marker that labels healthy gut stem cells also labels stem cells in benign intestinal tumors, which are precursors of cancer. In their latest study, his team engineered mice to carry a gene for a drug-inducible marker that once activated caused labeled cells to make molecules that fluoresce one of four colors. The experiment yielded single-color tumors consisting of several cell types, suggesting that each tumor originated from a single stem cell.
Cédric Blanpain and his group at the Free University of Brussels labeled individual tumor cells without targeting them specifically. They discovered that the cells showed two distinct patterns of division. They either produced a handful of cells before petering out, or went on to produce many cells. These results point to a distinct subset of cells acting as the engine of tumor growth. As the tumors became more aggressive, they were more likely to produce more stem cells that can divide indefinitely and less likely to produce differentiated cells, which can only divide a limited number of times. This could be the key to halting tumor development early, states Blanpain. Rather than eradicating cancer stem cells, therapies could try and make them differentiate into non-dividing cells.
These three papers clearly produce experimental evidence that cancer stem cells do exist, and that cancer cells act in more complex ways that previously thought. The next step is to figure out how the cells tracked in these experiments relate to putative cancer stem cells identified by years of transplantation studies.
References:
“Defining the mode of tumour growth by clonal analysis” by Gregory Driessens, Benjamin Beck, Amélie Caauwe, Benjamin D. Simons and Cédric Blanpain, 1 August 2012, Nature.
DOI: 10.1038/nature11344
“A restricted cell population propagates glioblastoma growth after chemotherapy” by Jian Chen, Yanjiao Li, Tzong-Shiue Yu, Renée M. McKay, Dennis K. Burns, Steven G. Kernie and Luis F. Parada, 1 August 2012, Nature.
DOI: 10.1038/nature11287
“Lineage Tracing Reveals Lgr5+ Stem Cell Activity in Mouse Intestinal Adenomas” by Arnout G. Schepers, Hugo J. Snippert, Daniel E. Stange, Maaike van den Born, Johan H. van Es, Marc van de Wetering and Hans Clevers, 1 August 2022, Science.
DOI: 10.1126/science.1224676
Never miss a breakthrough: Join the SciTechDaily newsletter.
Follow us on Google and Google News.