A gene responsible for prenatal death when critical transgenerational instructions are missing from egg cells has been discovered by researchers at the RIKEN Center for Integrative Medical Sciences (IMS) in Japan, led by Azusa Inoue.
“This study identified genes critical for fetal development whose expression is controlled by histone modifications transmitted from eggs to the next generation,” says Inoue. “The findings have implications for understanding infertility and developing treatments.”
Genomic Imprinting and Transgenerational Epigenetics
In order for embryos to develop normally, egg and sperm cells must first acquire critical biological instructions before they meet up. When an egg is fertilized, some of these instructions inform genes whether they should be switched on or off, depending on whether they originated from the mother or the father. This process is known as genomic imprinting, and it is the subject of the new research study.
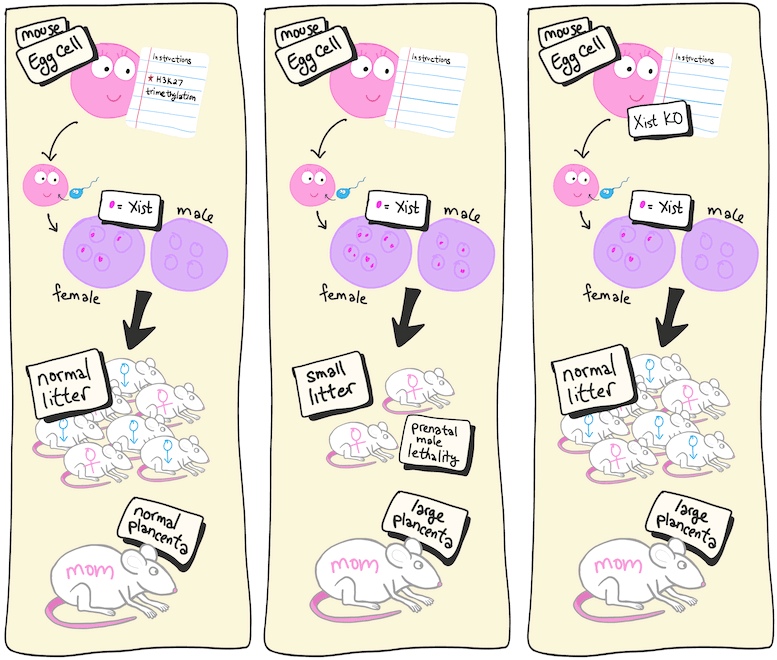
When modifications in gene expression are passed on to the next generation, they are called transgenerational epigenetic changes because they’re inheritable changes even though the DNA code remains unchanged. Inoue and his team have been studying a specific set of transgenerational epigenetic instructions given to egg cells called histone H3 lysine 27 (H3K27) trimethylation. In previous studies, they found that preventing these instructions led to prenatal death, particularly for male embryos, and also to enlarged placentas in the mothers. The new study asked whether those outcomes were directly related to failed imprinting.
Investigating the Xist Gene and Its Connection to Prenatal Death
The study began by knocking out a gene required for H3K27 trimethylation in eggs so that the transgenerational instructions could not be given. Next, the team added a knockout of the Xist gene to these eggs. Because the male offspring tended to die, the researchers suspected that the culprit was a gene on the sex chromosome. As it turns out, there are nine maternal genes known to be suppressed in embryos in favor of the ones with paternal origins. And only one, Xist, is on the X-chromosome.
The results were almost as expected. Prenatal death was greatly reduced, and the male-skewed lethality was gone after knocking out Xist. This showed that failed Xist imprinting was the reason for the prenatal death. However, the placenta was still enlarged. Reasoning that this was likely related to excess expression of the other eight genes that failed to imprint, the team created eight different deletion mutants in the double knockout embryos. They found that for three of the genes, this resulted in normal-sized placentas.
“We succeeded in curing developmental defects in a mouse model that otherwise suffers from prenatal lethality and placental malformation due to the lack of transgenerational epigenetic instructions from mothers,” says Inoue. The researchers plan to conduct more experiments to determine how these specific biological instructions are established when egg cells are created, and whether environmental factors can influence the process.
Reference: “Noncanonical imprinting sustains embryonic development and restrains placental overgrowth” by Shogo Matoba, Chisayo Kozuka, Kento Miura, Kimiko Inoue, Mami Kumon, Ryoya Hayashi, Tatsuya Ohhata, Atsuo Ogura and Azusa Inoue, 28 April 2022, Genes & Development.
DOI: 10.1101/gad.349390.122
Never miss a breakthrough: Join the SciTechDaily newsletter.
Follow us on Google and Google News.
2 Comments
The lethal malformation òf life defining cells comming in genes from parents can be erased out and replaced by normal useful cells by successive or simultaneous processes of gene treatement.Environmentplays an equal role in elemination of fetal genes or transplantation of healthy genes.
Substraction of fetal negative genes appeared from inheritation and addition of genes as required is possible,but environment plays an equal role.