
According to data from the Ministry of Land, Infrastructure, and Transport of Korea, there were approximately 30,000 hydrogen-powered vehicles registered by 2022, representing a threefold increase compared to 2018. However, the country only has 135 hydrogen fueling stations.
For hydrogen to be more accessible for vehicles and to be recognized as a reliable energy alternative, it’s essential to bring down the production costs of hydrogen, ensuring it’s economically viable. Central to this objective is optimizing the efficiency of the electrolysis-hydrogen evolution process, which produces hydrogen from water.
Breakthrough in Hydrogen Production
Recently, a team of researchers comprising Professor In Su Lee, Research Professor Soumen Dutta, and Byeong Su Gu from the Department of Chemistry at Pohang University of Science and Technology (POSTECH) achieved a significant improvement in the production efficiency of hydrogen, a green energy source, through the development of a platinum nanocatalyst.

They accomplished this feat by depositing two different metals in a stepwise manner. The findings of their research were published in Angewandte Chemie, the esteemed journal that focuses on the field of chemistry.
Challenges and Innovation in Catalyst Development
Depositing distinct materials selectively on specific locations of a catalyst surface, whose size is in the nanometer range, poses substantial challenges. Unintended depositions may block the catalyst’s active sites or interfere with each other’s functions. This predicament has prevented the simultaneous deposition of nickel and palladium onto a single material. Nickel is responsible for activating water splitting while palladium facilitates the conversion of hydrogen ions into hydrogen molecules.
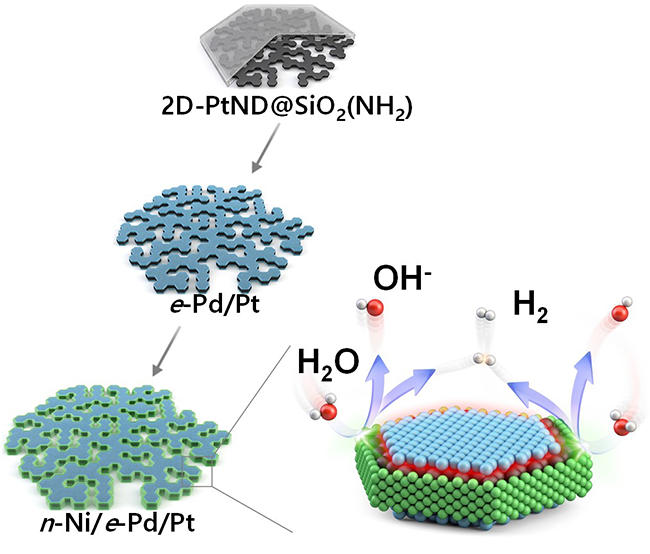
The research team developed a novel nanoreactor to finely control the location of metals deposited onto a 2D flat nanocrystal. Additionally, they devised a nano-scaled fine deposition process, enabling the coverage of different facets of the 2D platinum nanocrystal with different materials. This new approach led to the development of a “platinum-nickel-palladium” three-metal hybrid catalyst material achieved through consecutive depositions that selectively cover the flat surface and the edge of the 2D platinum nanocrystal with palladium and nickel nano thin films respectively.
Hybrid Catalyst’s Enhanced Efficiency
The hybrid catalyst featured distinct nickel/platinum and palladium/platinum interfaces positioned to facilitate the water splitting and hydrogen molecule generation processes respectively. Consequently, the collaborative occurrence of these two different processes significantly boosted the effectiveness of electrolysis-hydrogen evolution.
The research outcomes revealed that the three-metal hybrid nano catalyst exhibited a 7.9-fold increase in catalytic activity compared to the conventional platinum-carbon catalyst. Moreover, the novel catalyst demonstrated significant stability, maintaining its high catalytic activity even after a prolonged 50-hour reaction time. This resolved the issue of functional interferences or collisions between heterointerfaces.
Closing Remarks from the Research Team
Professor In Su Lee who led the research expressed his optimism by stating, “We have successfully developed harmonious heterointerfaces formed on a hybrid material, overcoming the challenges of the process.” He further added, “I hope the research findings will find widespread application in the development of catalytic materials optimized for hydrogen reactions.”
Reference: “Harmonious Heterointerfaces Formed on 2D-Pt Nanodendrites by Facet-Respective Stepwise Metal Deposition for Enhanced Hydrogen Evolution Reaction” by Byeong Su Gu, Soumen Dutta, Yu-Rim Hong, Odongo Francis Ngome Okello, Hyeonae Im, Seungil Ahn, Si-Young Choi, Jeong Woo Han, Sunmin Ryu and In Su Lee, 19 June 2023, Angewandte Chemie International Edition.
DOI: 10.1002/anie.202307816
The study was conducted with the support from the Leading Researcher Program of the National Research Foundation of Korea.
Never miss a breakthrough: Join the SciTechDaily newsletter.
Follow us on Google and Google News.
7 Comments
Thanks
Repeat after me and remember: hydrogen is not an energy *source* (as this article claims it is). In the big picture of energy production and use, hydrogen is an energy *storage medium*.
Wrong
Hmm. What grade did you get in your high school basic chemistry course?
Wow….and what do you want to power the process with? Oh, that’s right….coal and oil. What will you power with it?!?!? Oh, that’s right…..there is nothing at all that is powered by hydrogen. STFU and stop wasting our money in this BS.
I think I hydrogen proved we can create water hello….like making it rain using hydrogen to liquefy atmospheres … splitting water on a molecule level that’s titts.
To the Marxist environmental crusaders splitting water into hydrogen and oxygen is a threat to their agenda. They already got hoards to believe their big lie about carbon being at a dangerous level in atmosphere even though plants would prefer 4x as much. While complaining about energy input into splitting water, I presume they are in denial about infrastructure for charging electric cars, and THAT input requirement. They ignore feedback from firefighters and insurance companies as well. Marxist agendas seem more and more to be about creating chaos. Some of these imbeciles don’t even realize they are just dupes in a Marxist agenda to create chaos. They really are dumb enough to argue against hydrogen cracking even at 7 times improved efficiency! Their agenda is sacred. Science does not matter to them. Their warped political agenda to create chaos knows no boundaries.