
A new method uses microwaves to lower the energy required for certain industrial processes.
Some industrial chemical production processes depend on heat, but traditional heating methods are often wasteful because they warm large areas that do not actually need it. A research team including scientists from the University of Tokyo developed a method to focus heat only where it is needed.
Their approach uses microwaves, similar to those in a household microwave oven, to excite specific elements within the target materials. The new system achieved energy efficiencies about 4.5 times higher than conventional techniques.
A green transformation approach to industrial chemistry
Although climate change involves more than just energy production and carbon dioxide (CO2), lowering energy demand and emissions remains a key challenge for science and engineering. Under the broader goal of green transformation, Lecturer Fuminao Kishimoto and his colleagues in the Department of Chemical System Engineering at the University of Tokyo are developing cleaner, more efficient industrial methods.
Their latest work could improve processes used in chemical synthesis and may lead to other environmental benefits. The basic idea behind their innovation is surprisingly simple.
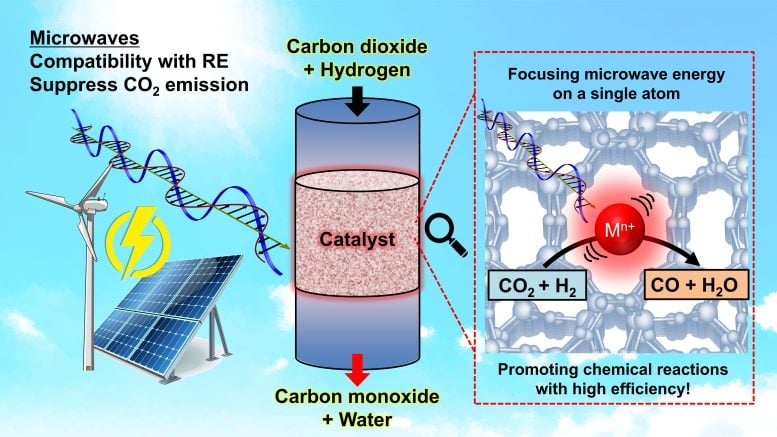
“In most cases, chemical reactions occur only at very small, localized regions involving just a few atoms or molecules. This means that even within a large chemical reactor, only limited parts truly require energy input for the reaction,” said Kishimoto. “However, conventional heating methods, such as combustion or hot fluids, disperse thermal energy throughout the entire reactor. We started this research with the idea that microwaves could concentrate energy on a single atomic active site, a little like how a microwave oven heats food.”
How it works: tuning microwaves for precision heating
As Kishimoto explained, the concept resembles that of a microwave oven but operates under different conditions. Instead of targeting polar water molecules at roughly 2.45 gigahertz (a frequency also used by many Wi-Fi signals, which explains why internet connections can sometimes falter when reheating food), the researchers tuned their microwaves to about 900 megahertz. This lower frequency proved optimal for exciting the material they were studying—zeolite—a porous substance that can efficiently absorb and transfer heat.
“The most challenging aspect was proving that only a single atomic active site was being heated by the microwaves. To achieve this, we spent four years developing a specialized experimental environment at Japan’s world-class large synchrotron radiation facility, SPring-8,” said Kishimoto.
“This involved using spongelike zeolite, which is ideal because we can control the sizes of the sponge cavities, allowing us to balance different factors of the reactions. Inside the sponge cavities, indium ions act like antennas. These are excited by the microwaves which creates heat, which can then be transferred to reaction materials passing through the sponge.”
Applications in fuel creation and carbon recycling
By selectively delivering heat to specific materials, lower overall temperatures can be used to achieve reactions which are otherwise very demanding, such as water decomposition or methane conversion, both useful to create fuel products. They can further improve selectivity by varying the pore size of the zeolite sponge, with smaller pores yielding greater efficiency and larger pores enabling greater control over reactions.
And one key advantage is that this technique can even be used in carbon capture, recycling CO2 as part of the methane conversion, and even recycling plastics more easily.

The challenge now will be in how to scale this up to encourage industrial adoption — things that work in the lab don’t directly translate into large industrial settings easily. And there are some limitations to the research that would also need to be addressed first.
The material requirements are quite complex and aren’t simple or cheap to produce; it’s hard to precisely measure temperatures at the atomic scale, so current data rely on indirect evidence, and more direct means would be preferred. And, despite the improvements in efficiency, there is still room for improvement here too as there are heat and electrical losses along the way.
“We aim to expand this concept to other important chemical reactions beyond CO2 conversion and to further optimize catalyst design to improve durability and scalability. The technology is still at the laboratory stage. Scaling up will require further development of catalysts, reactor design and integration with renewable power sources,” said Kishimoto.
“While it is difficult to give an exact timeline, we expect pilot-scale demonstrations within the next decade, with broader industrial adoption depending on progress in both technology and energy infrastructure. To achieve this, we are seeking corporate partners to engage in joint development.”
Reference: “Focused thermal energy at atomic microwave antenna sites for ecocatalysis” by Ryo Ishibashi, Fuminao Kishimoto, Tatsushi Yoshioka, Hiroki Yamada, Koki Muraoka, Toshiaki Ina, Hiroki Taniguchi, Akira Nakayama, Toru Wakihara and Kazuhiro Takanabe, 10 October 2025, Science Advances.
DOI: 10.1126/sciadv.ady4043
Funding: F.K. acknowledges that this work was supported by Japan Society for the Promotion of Science (JSPS) KAKENHI Grant-in-Aid for Transformative Research Areas (A) (21H05550 and 23H04097) and Grant-in-Aid for Scientific Research (B) (24K01254). K.M. acknowledges the support of JST PRESTO (JPMJPR2378). T.Y. acknowledges a research fellow (DC1) of Japan Society for the Promotion of Science (JSPS) (22KJ0675).
Never miss a breakthrough: Join the SciTechDaily newsletter.
Follow us on Google and Google News.
1 Comment
thqnks for this