Researchers have developed a new catalyst that could make hydrogen production cheaper and more practical for renewable energy systems.
Hydrogen has long been viewed as one of the most promising clean fuels because it can store renewable energy and produce no carbon emissions when used.
Yet producing hydrogen efficiently and affordably has remained a major challenge, largely because today’s most effective electrolyzers rely on scarce and expensive platinum-group metals. Finding cheaper alternatives that can still deliver high performance is considered a key step toward making large-scale renewable energy storage practical.
Now, a research team led by Gang Wu, professor of energy, environmental & chemical engineering in the McKelvey School of Engineering at Washington University in St. Louis, has developed a new catalyst that could help overcome those barriers.
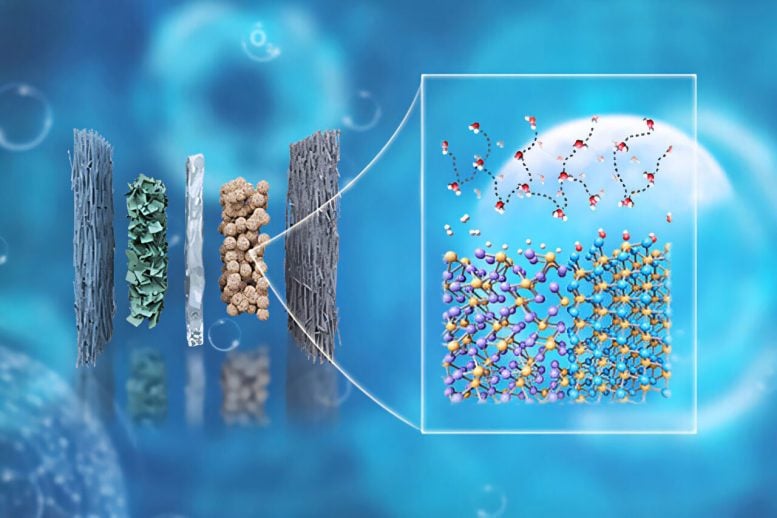
The team created a heterostructure catalyst for an anion-exchange membrane water electrolyzer (AEMWE), a system that uses renewable electricity to split water into hydrogen and oxygen. By combining two phosphide materials, the researchers designed a catalyst that produces hydrogen efficiently without relying on costly platinum-based materials, potentially lowering the price of clean hydrogen production.
Cheaper catalysts could store energy
Wu’s team has been searching for substitutes for catalysts that depend on expensive platinum group metals. Their approach centers on using electricity from sunlight, wind, or water to separate hydrogen from water, turning renewable power into a fuel that can be stored and used later.
“Going from water to hydrogen is a very desirable way we are able to store energy for different applications,” Wu said. “Hydrogen itself can be used as an energy carrier and is useful for different chemical industries and manufacturing.”
Two phosphides improve hydrogen extraction
The team combined rhenium phosphide (Re2P) with molybdenum phosphide (MoP) to form a composite catalyst whose parts work together to improve hydrogen production. Rhenium helps with hydrogen adsorption and desorption, while molybdenum helps accelerate water splitting, which supplies protons in the alkaline electrolyte.
When the catalyst was paired with a nickel-iron anode, the resulting cathode performed better than a leading cathode made from other materials and also surpassed a PGM benchmark. The researchers also showed that it could run at industrial current densities of 1 and 2 amperes per square centimeter for more than 1,000 hours. Wu said this makes it one of the most durable cathodes free of platinum group metals for anion-exchange membrane water electrolyzers.
“Our findings allowed us to rationalize the critical role of engineering the hydrogen-bond network at the catalyst/electrolyte interface in designing high-efficiency, low-cost AEMWEs,” Wu said. “Our catalyst showed the lowest resistance across the studied potential range, which suggests the fastest hydrogen adsorption kinetics among the studied catalysts. This newly achieved performance and durability metrics make our catalyst one of the most promising membrane electrode assemblies for practical anion-exchange membrane water electrolyzers.”
The experiments were conducted at lab scale, but the team now plans to study whether the cathode can be used at industrial scale.
Reference: “Designing a Dry Cathode via Hydrogen-Bond Network Regulation at Phosphide Heterostructure/Electrolyte Interfaces for Alkaline Water Electrolysis” by Jiashun Liang, Yu Li, Chun-Wai Chang, Mingxuan Qiao, Zhenxing Feng, Chaochao Dun, Wan-Lu Li and Gang Wu, 7 April 2026, Journal of the American Chemical Society.
DOI: 10.1021/jacs.6c02768
This work was financially supported by G. Wu’s startup fund at Washington University in St. Louis.
Never miss a breakthrough: Join the SciTechDaily newsletter.
Follow us on Google and Google News.
7 Comments
thanks for this
Cheaper and more efficient is an improvement but the fundamental fact remains….it takes more energy to split water into H and O2 than you get back later using it as a fuel. You can’t cheat physics.
We are facing a global drought and you are taking water out of the water cycle, separating into a state that will never be water again. I hope you are at least using sea water.
Wtf are yout alking about 70% of planet surface is water and hydrogen can be recombined with oxygen back into water, like what are you even on about?!?!?!?!?!
Errrr. When they burn the hydrogen, hey presto! It turns back into water. No loss of water.
When you use hydrogen as a fuel, by burning it up or generating electricity in a fuel cell, it combines with oxygen and becomes water again. Also if you combine it with carbon, to generate synthetic methane or ethanol, when you burn those, the hydrogen will combine with oxygen and become water again.
There are other stable molecules where hydrogen could be sequestered, but as a fuel it almost always become water again while shedding out the energy that was used to separate it from oxygen in the first place.
Calgon! An ancient Chinese secret!
Edit: This is a quote from a movie