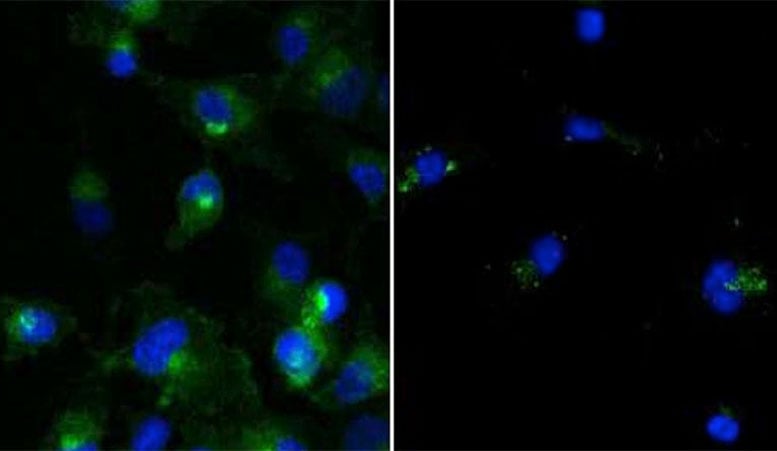
Cells treated (right) or untreated (left) with a PROTAC that degrades the target protein (green).
Two new research papers from Yale University reinforce the benefits of a novel therapy that hijacks the cell’s own protein degradation machinery to destroy cancer cells. The Yale researchers report the findings in the journal Cell Chemical Biology.
The new approach to drug discovery, called Proteolysis Targeting Chimeras or PROTACs, was developed in the laboratory of Craig Crews, the Lewis B. Cullman Professor of Molecular, Cellular, and Developmental Biology, professor of chemistry and pharmacology, as well as executive director of the Yale Center for Molecular Discovery.
The system engages the cell’s own protein degradation machinery to destroy targeted proteins by tagging them for removal. Most drugs are based on the ability of small molecules to bind to and block the function of disease-causing proteins, but some proteins are resistant to such intervention.
“This system will help us change the current small-molecule drug paradigm that fails to target 75% of rogue proteins,” said Crews, scientific founder of Arvinas LLC, the New Haven biotechnology company developing the concept.
The first paper shows for the first time that PROTAC system can target mutant RTK proteins, which have been linked to several forms of cancer. The second paper proves that the PROTAC system can target rogue proteins with greater specificity than traditional approaches.
Yale’s George M. Burslem and Blake E. Smith are first authors of the first paper. Smith and Yale’s Daniel P. Bondeson are co-first authors of the second paper.
The two papers were primarily funded by the National Institutes of Health. Crews is a shareholder of Arvinas, which also provided researchers to the projects.
References:
- “The Advantages of Targeted Protein Degradation Over Inhibition: An RTK Case Study” by George M. Burslem, Blake E. Smith, Ashton C. Lai, Saul Jaime-Figueroa, Daniel C. McQuaid, Daniel P. Bondeson, Momar Toure, Hanqing Dong, Yimin Qian, Jing Wang, Andrew P. Crew, John Hines and Craig M. Crews, 9 November 2017, Cell Chemical Biology.
DOI: 10.1016/j.chembiol.2017.09.009 - “Lessons in PROTAC Design from Selective Degradation with a Promiscuous Warhead” by Daniel P. Bondeson, Blake E. Smith, George M. Burslem, Alexandru D. Buhimschi, John Hines, Saul Jaime-Figueroa, Jing Wang, Brian D. Hamman, Alexey Ishchenko and Craig M. Crews, 9 November 2017, Cell Chemical Biology.
DOI: 10.1016/j.chembiol.2017.09.010






Be the first to comment on "New Research Shows Cellular Clean-Up Can Also Sweep Away Forms of Cancer"