
Researchers have discovered a strong correlation between the development of schizophrenia and a rare form of a genetic variant known as tandem repeats.
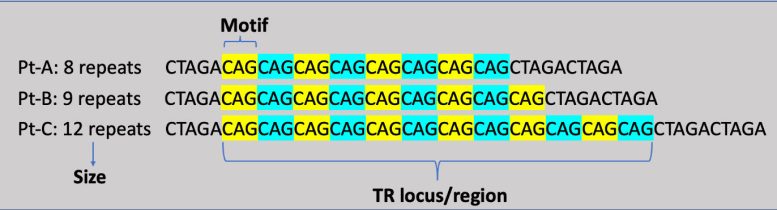
In our cells, DNA acts as a language, creating the unique characteristics that make each of us who we are. Tandem repeats occur within DNA when a pattern of one or more nucleotides, the building blocks of DNA made up of the chemicals cytosine (C), adenine (A), guanine (G), and thymine (T), is repeated multiple times in a row. An example of this could be: CAG CAG CAG, in which the sequence CAG is repeated three times.
The University of North Carolina School of Medicine lab of Jin Szatkiewicz, Ph.D., associate professor of genetics, and colleagues, have conducted one of the most extensive investigations of tandem repeats in schizophrenia, using cutting-edge whole-genome sequencing and machine learning techniques. Their research has shed light on the role of tandem repeats in the development of this debilitating disease.
Findings on Tandem Repeats in Schizophrenia
Published in the journal Molecular Psychiatry, the research shows that individuals with schizophrenia had a significantly higher rate of rare tandem repeats in their genomes – 7% more than individuals without schizophrenia. And they observed that the tandem repeats were not randomly located throughout the genome; they were primarily found in genes crucial to brain function and known to be important in schizophrenia, according to previous studies.
“We think this discovery opens doors for future functional studies on the precise biological mechanism of such variants,” said Szatkiewicz, who is also an adjunct assistant professor of psychiatry. “Understanding the biological cause of schizophrenia will enable future development of diagnostic tests, effective pharmaceuticals, and personalized treatments.”
Tandem repeats usually don’t have negative health implications. However, based on the location of tandem repeats in the genome and how long they are, they can contribute to disease. For example, Huntington’s disease is caused by a tandem repeat in the HTT gene that has been abnormally expanded. The onset of the disease will happen once the sequence of cytosine-adenine-guanine (CAG) repeats more than 36 times on the HTT gene. The longer repeat expansions lead to abnormal protein products with an extended track of glutamine that is toxic to brain cells. These repeats are inherited and tend to grow longer and longer in successive generations with increasing disease severity or decreasing age of onset.
In their current study, Szatkiewicz and her team looked at the entire genomes of 2,100 individuals to find tandem repeats that looked abnormally long and were unique or rare. Because all participants provided access to their medical records, the team was able to compare these long and rare repeat DNA sequence samples from people who had schizophrenia versus samples from people in the study who didn’t. This allowed the researchers to determine which of these tandem repeats may be involved with the development of schizophrenia.
Functional Insights into Schizophrenia
Using gene network analysis, the authors of this study demonstrated that genes with rare tandem repeats found in schizophrenia primarily impact synaptic and neuronal signaling functions. In addition, these genes are highly evolutionarily conserved, indicating important biological functions and therefore the significant impact that tandem repeats might exert.
The UNC School of Medicine researchers then collaborated with scientists from The Hospital for Sick Children in Toronto to see if this increased level of rare tandem variants would also be found in another independently collected group of samples. The Szatkiewicz findings were replicated in the Canadian investigation, indicating that this newly discovered link between tandem repeats and schizophrenia is quite strong.
“We think this is an important study,” said Szatkiewicz. “We’re confident our work sheds significant light on the role of tandem repeat DNA mutations play in the development of schizophrenia.”
Reference: “Rare tandem repeat expansions associate with genes involved in synaptic and neuronal signaling functions in schizophrenia” by Jia Wen, Brett Trost, Worrawat Engchuan, Matthew Halvorsen, Linda M. Pallotto, Aleksandra Mitina, NaEshia Ancalade, Martilias Farrell, Ian Backstrom, Keyi Guo, Giovanna Pellecchia, Bhooma Thiruvahindrapuram, Paola Giusti-Rodriguez, Jonathan David Rosen, Yun Li, Hyejung Won, Patrik K. E. Magnusson, Ulf Gyllensten, Anne S. Bassett, Christina M. Hultman, Patrick F. Sullivan, Ryan K. C. Yuen and Jin P. Szatkiewicz, 16 November 2022, Molecular Psychiatry.
DOI: 10.1038/s41380-022-01857-4
This research was primarily funded through grants from the National Institute of Mental Health and the SciLifeLab National Project.
Never miss a breakthrough: Join the SciTechDaily newsletter.
Follow us on Google and Google News.
1 Comment
What purpose is served by the use of horror movie-type imagery to accompany articles about schizophrenia? These images convey nothing about the disease, and they only serve to stigmatize those suffering from it. Or, worse, images like those used for this article may induce an unjustified fear of schizophrenics.