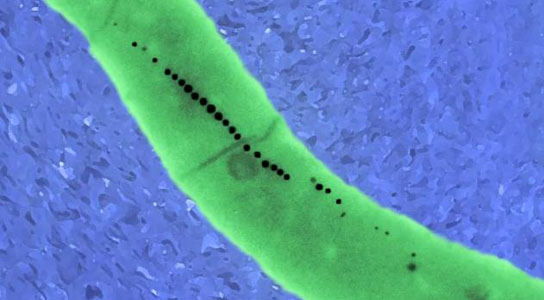
To better understand the underlying biology of induced magnetization, researchers from the Wyss Institute for Biologically Inspired Engineering at Harvard University and Harvard Medical School have induced magnetism to a non-magnetic organism. While growing yeast, the researchers introduced one protein, ferritin, and knocked down the expression of another protein, ccc1, which resulted in cells with approximately three-fold higher levels of magnetism than wild-types supplemented with iron.
Magnetic fields span the globe, but few organisms can sense them. In fact, how these few magnetically-aware organisms, such as butterflies and bees, gain their magnetism remains one of biology’s unsolved mysteries.
Now, using the tools of synthetic biology, researchers from the Wyss Institute for Biologically Inspired Engineering at Harvard University and Harvard Medical School have induced magnetism to a non-magnetic organism: yeast. And because they used a common cell pathway, the findings suggests that such magnetism could be achieved in many types of cells for a variety of industrial, medical and research applications. The findings appear in the February 28 issue of PLoS Biology.
“Magnetism in nature is a unique and mysterious biological function that very few living systems exploit,” said principal investigator Pam Silver, the Elliott T. And Onie H. Adams Professor of Biochemistry and Systems Biology at HMS. “So while magnetic yeast may sound like a curiosity, it’s actually a highly significant first step toward harnessing this natural phenomenon and applying it to all sorts of important practical purposes.”
Silver’s team took an engineering approach. First author Keiji Nishida, research fellow in systems biology at HMS, first grew yeast in a medium containing iron. The yeast cells took in the element and stored it inside cellular containers called vacuoles. By placing a magnet under the culture, Nishida saw that the yeast cells had become slightly magnetic.
“We used the design, build, test cycle of engineers,” said Silver, who is also a core faculty member at the Wyss Institute. “It worked, but we wanted to make it better. That’s where we employ synthetic biology.”
The tools of synthetic biology manipulate natural biological instructions, such as genes or cellular signals. Since the instructions that make up the resulting biological organism are not found together in nature, the new biology is considered “synthetic.”
To improve the magnetized yeast, Nishida employed two tools of synthetic biology: He introduced one protein, ferritin, which joins with iron and prevents it from becoming toxic to the cell. He also knocked down the expression of another protein, ccc1, which carries the iron into the cell’s vacuoles. “The cells we built ended up forming magnetic iron crystals in their mitochondria, not a place yeast normally put iron,” Nishida said. They also exhibited approximately three-fold higher levels of magnetism than wild-types supplemented with iron.
Silver and Nishida also searched for genes that might contribute to magnetism. They screened collections of yeast with different genes knocked out for mutants that affect magnetism. They focused on iron homeostasis genes, which regulate how cells store iron, and oxidative stress genes, which regulate how cells use iron.
They zeroed in on one gene: TCO89. Increased levels dialed up magnetism, while decreased levels dialed it down. Further experiments showed that, to achieve increased magnetism, the TCO89 protein must interact with TORC1, a protein complex that regulates cell metabolism. TORC1 is highly conserved, meaning its form and function is similar from yeast to more advanced, and even human cells. “This suggests that other cells might potentially be similarly magnetized,” Silver said.
Such a capability could have many applications. In an industrial setting, magnetization could be a means of isolating cells during a process. “There’s often a need to suck out cells that are making a product or contaminating a bioprocessor,” Silver said.
Tissue engineers could guide cells magnetically to layer themselves on a scaffold. And with the application of additional synthetic biology tools, clinicians may one day use cells engineered to respond to a magnetic field by growing or healing, or implant magnetic stem cells that can be tracked with magnetic resonance imaging.
“This study is an example of the facility with which you can engineer biology,” said Silver, who imagines that a similar approach could be used to mimic other unusual natural phenomena. “Biology is where organic chemistry was about 80 years ago. Now we know enough about biology that it could take over from chemistry.”
The synthetic yeast may also aid in the study of magnetism in nature. Although Nishida and Silver may not be able to study naturally magnetic bacteria directly — it doesn’t do well in the lab — “we have our own private evolvable system,” Silver said. “It may or may not be artificial, but it is a starting point that allows us to study the evolvability of the phenomenon.”
Reference: “Making Magnetic Yeast” by Stephanie Huang, 28 February 2012, PLoS Biology.
DOI: 10.1371/journal.pbio.1001274
Never miss a breakthrough: Join the SciTechDaily newsletter.
Follow us on Google and Google News.
1 Comment
Magnetic Manipulation: Biology and Biomimetic Materials