Researchers at the Max Planck Institute have now clarified exactly how hydroxyl radicals, which oxidize volatile organic compounds such as methane and isoprene, are recycled once they have finished their chemical cleaning duties. The research shows that isoprene, naturally released by plants, contributes to the reduction as well as to the formation of OH radicals and recycling efficiency of OH radicals depends on isoprene quantity.
The atmosphere’s robust capacity to clean itself is owed to the consistent recycling of its cleaning agent. Scientists from the Max Planck Institute for Chemistry in Mainz have now clarified exactly how hydroxyl radicals, which break up organic compounds in the air, are recycled. According to their findings, the reactive molecules can be produced when isoprene is broken down. Isoprene is released into the atmosphere by plants and was, until now, only known for using up hydroxyl radicals during its chemical cleaning. However, it appears that at low hydroxyl concentrations, more of the atmospheric cleaning agent is produced than is removed. Therefore, isoprene is acting as a sort of buffer, which can mitigate the increase in greenhouse gases and other air pollutants.
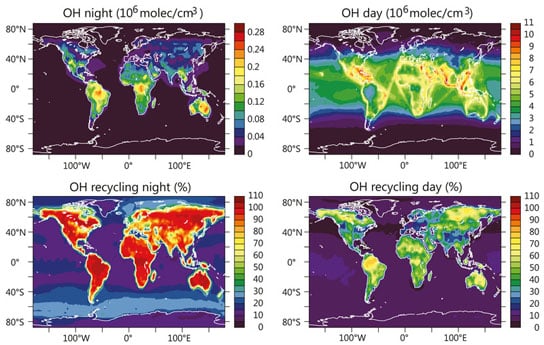
Billions of tonnes of natural and anthropogenic gases are emitted into the Earth’s atmosphere every year. If these gases were not removed by chemical reactions, global warming would be much greater and the air quality much poorer. The most important cleaning agents in the atmosphere are hydroxyl (OH) radicals, which oxidize volatile organic compounds such as methane and isoprene. The reaction with the short-lived but highly reactive molecule transforms these gases into water-soluble compounds that can be removed from the atmosphere by precipitation. However, the oxidation also leads to the formation of ozone and aerosol particles, which in turn affect air quality and the regional climate.
Measurements of OH radical concentrations above tropical rainforests suggest that OH radicals are recycled once they have finished their chemical cleaning duties. This is why the atmosphere’s self-cleaning capacity has proved to be relatively stable in the face of air pollution. Domenico Taraborrelli and his fellow scientists at the Max Planck Institute for Chemistry in Mainz have now clarified how a certain recycling mechanism for the hydroxyl radicals influences the OH balance in the atmosphere. What plays the key role here is a substance that was previously known only for consuming hydroxyl radicals as it broke down. The substance in question is isoprene. The volatile organic compound is naturally released into the atmosphere in large quantities by plants and, together with other chemically-related terpenes, forms one of the main components in the essential oils that are important in many plant odors. It is estimated that vegetation produces about 500 million tonnes of isoprene annually, with the majority produced during the day in tropical rainforests.
The hydroxyl concentration determines how isoprene is broken down
“We discovered that the system is multifaceted, because the oxidation of isoprene contributes to the reduction as well as to the formation of OH radicals, so that it both sustains and limits itself,” said Domenico Taraborrelli, lead author of a new study. “The recycling efficiency of OH radicals therefore depends on their quantity.” If the OH concentration is high, little is recycled; if it is low, a lot is produced.
“This buffering effect explains why much higher concentrations of hydroxyls are measured over rainforests than the atmospheric chemistry models suggested,” said Domenico Taraborrelli. He and his fellow scientists resolved this inconsistency by adding complex reaction cascades to a model of global atmospheric chemistry. In the model, OH radicals first convert isoprene into a hydro-peroxy-aldehyde (HPAL). What happens next depends on how much of the OH radical is in the air. If the concentration is low, the HPAL reacts with molecular atmospheric oxygen in a chain of reactions kicked off by sunlight. This ultimately results in more OH radical than was consumed by the initial reaction which formed an HPAL. If the air contains a large concentration of OH radicals, the amount of HPAL is reduced even further. The OH concentration therefore falls.
“For the interaction between biosphere and atmosphere, the buffering of the OH radical quantity is very important, since it provides large forests with the ability to maintain their self-cleaning capacity,” explains Domenico Taraborelli. The Mainz-based atmospheric chemists even suspect that volatile organic compounds released as a result of human activity could buffer the atmosphere’s self-cleaning capacity in the same way as isoprene does.
“Natural ecosystems are better buffered than we suspected”
The fact that the atmosphere retains its ability to clean itself in the face of air pollution is something that is likely to stand the global climate in good stead: many volatile organic compounds, like methane, exert a strong greenhouse effect; and in the course of climate change, they are likely to be released from natural sources in even greater quantities. However, the consequences of this could be less severe than we had assumed. That is because, in a warmer climate, plants are expected to release more isoprene into the atmosphere as well. And, according to the latest findings, a higher isoprene concentration leads to the production of more hydroxyl radicals, which clean the atmosphere of greenhouse gases. “Our results also indicate that increasing isoprene emissions, which we expect from global warming, will not enhance the climate effect,” concludes Jos Lelieveld, Director at the Max Planck Institute for Chemistry. “This shows that natural ecosystems and their atmospheric environment are better buffered than we suspected.”
The researchers in Mainz now want to study how the atmosphere’s self-cleaning capacity reacts to perturbations, such as sudden methane releases from permafrost soils. Global warming is causing major areas of permafrost in countries like Russia to thaw, which may release huge quantities of methane and accelerate the global warming effect.
Never miss a breakthrough: Join the SciTechDaily newsletter.
Follow us on Google and Google News.