Scientists have uncovered a hidden structure in one of our most mysterious senses.
Smell plays a vital role in daily life. It helps us detect danger, enriches the way food tastes, and is closely tied to memory and emotion. Even so, scientists have long struggled to fully explain how this sense works at a basic biological level.
“Olfaction is super-mysterious,” said Sandeep (Robert) Datta, professor of neurobiology in the Blavatnik Institute at Harvard Medical School. Compared with sight, hearing, and touch, the science behind smell has remained less developed.
First Detailed Map of Smell Receptors in the Nose
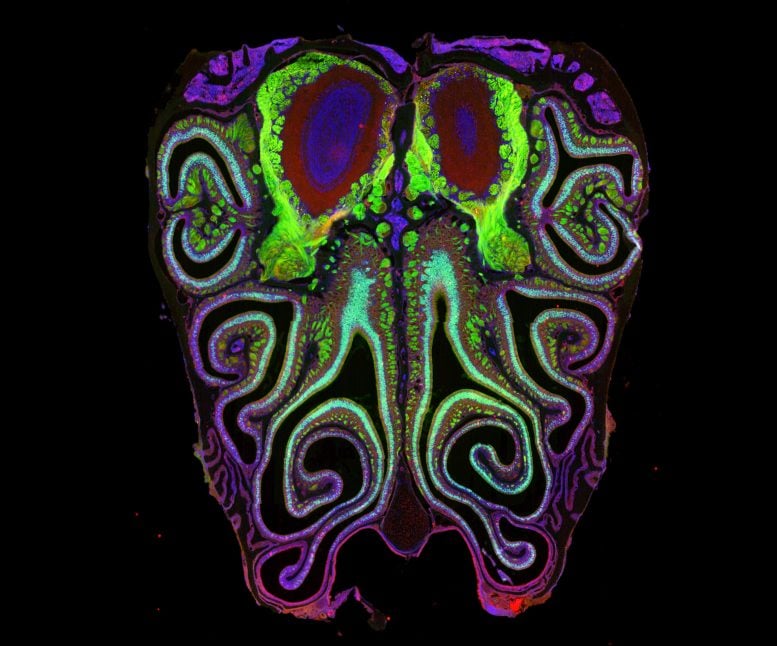
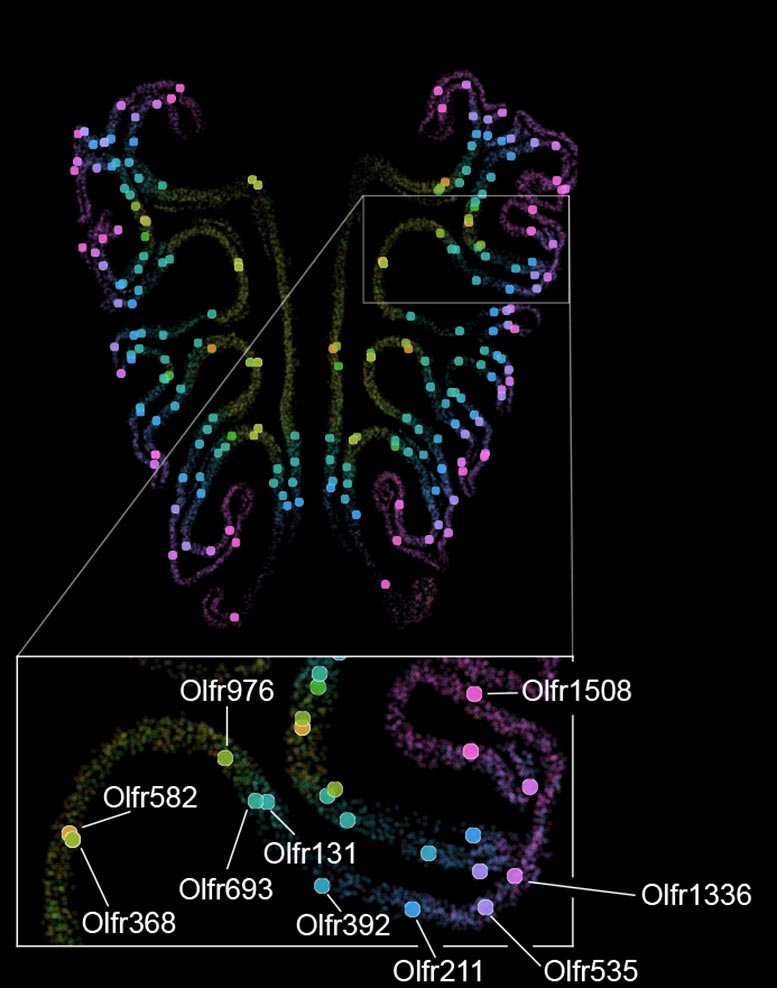
Using mice, Datta and his team have now produced the first detailed map showing how more than a thousand different smell receptors are arranged inside the nose.
Their findings overturn a long-standing assumption. Instead of being randomly distributed, the neurons that carry these receptors are arranged in a highly organized way. They form horizontal stripes that extend from the top of the nose to the bottom, grouped by receptor type.
“Our results bring order to a system that was previously thought to lack order, which changes conceptually how we think this works,” said Datta, senior author of the study.
The team also found that this layout in the nose aligns with corresponding maps in the olfactory bulb of the brain. This connection offers new clues about how scent signals travel from the nose into the brain.
The findings were published April 28 in Cell.
Why Smell Lacked a Map for So Long
Scientists have long known how sensory receptors are arranged in the eyes, ears, and skin, and how those arrangements connect to the brain. Smell has been the exception.
“Olfaction has been the one exception; it’s the sense that has been missing a map for the longest time,” Datta said.
Part of the challenge comes from the system’s complexity. Mice have around 20 million olfactory neurons that express more than a thousand types of smell receptors. In contrast, color vision depends on just three main receptor types. Each smell receptor is tuned to detect a specific group of odor molecules.
Researchers began identifying smell receptors in 1991. Over the next 35 years, studies attempted to determine whether a map existed. Early observations showed that receptors were limited to a few broad zones, leading scientists to believe their distribution was mostly random.
As new genetic tools advanced, Datta and his colleagues decided to revisit the question with more powerful techniques.
Mapping Millions of Neurons Reveals Hidden Order
In the new study, the researchers examined about 5.5 million neurons from more than 300 mice. They combined single-cell sequencing, which identifies the receptors expressed by each neuron, with spatial transcriptomics, which shows where those neurons are located.
“This is now arguably the most sequenced neural tissue ever, but we needed that scale of data in order to understand the system,” Datta said.
The analysis revealed a clear pattern. Neurons are arranged in tightly packed, overlapping horizontal stripes based on the type of smell receptor they express. This structure was consistent across animals and closely matched how smell information is organized in the brain, similar to patterns seen in vision, hearing, and touch.
Retinoic Acid Helps Shape the Smell Map
The researchers also explored how this precise arrangement forms. They identified retinoic acid, a molecule that regulates gene activity, as a key factor.
A gradient of retinoic acid within the nose appears to guide neurons, determining which receptor each one expresses based on its position. When the researchers increased or decreased levels of this molecule, the entire receptor map shifted up or down.
“We show that development can achieve this feat of organizing a thousand different smell receptors into an incredibly precise map that’s consistent across animals,” Datta said.
A separate study led by the lab of Catherine Dulac, the Xander University Professor in the Department of Molecular and Cellular Biology at Harvard University, that published in the same issue of Cell had consistent findings.
What the Discovery Means for Treating Loss of Smell
The discovery provides a foundation for understanding how smell works and could have important medical implications. Loss of smell currently has limited treatment options, despite its impact on safety, nutrition, and mental health.
“We cannot fix smell without understanding how it works on a basic level,” Datta said.
Researchers are now investigating why the receptor stripes appear in their specific order and whether similar patterns exist in humans. This work could guide future treatments such as stem cell therapies or brain-computer interfaces for restoring smell, as well as addressing related issues like increased risk of depression.
“Smell has a really profound and pervasive effect on human health, so restoring it is not just for pleasure and safety but also for psychological well-being,” Datta said. “Without understanding this map, we’re doomed to fail in developing new treatments.”
Reference: “A spatial code governs olfactory receptor choice and aligns sensory maps in the nose and brain” by David H. Brann, Tatsuya Tsukahara, Cyrus Tau, Dennis Kalloor, Rylin Lubash, Lakshanyaa Thamarai Kannan, Nell Klimpert, Mihaly Kollo, Martín Escamilla-Del-Arenal, Bogdan Bintu, Andreas Schaefer, Alexander Fleischmann, Thomas Bozza and Sandeep Robert Datta, 28 April 2026, Cell.
DOI: 10.1016/j.cell.2026.03.051
Additional authors on the paper include David Brann, Tatsuya Tsukahara, Cyrus Tau, Dennis Kalloor, Rylin Lubash, Lakshanyaa Kannan, Nell Klimpert, Mihaly Kollo, Martin Escamilla-Del-Arenal, Bogdan Bintu, Andreas Schaefer, Alexander Fleischmann, and Thomas Bozza.
Funding for the research was provided by the National Institutes of Health (grants R01DC021669, R01DC021422, R01DC021965, and F31DC019017), the Yang Tan Collective at Harvard, and a National Science Foundation Graduate Research Fellowship.
Never miss a breakthrough: Join the SciTechDaily newsletter.
Follow us on Google and Google News.
1 Comment
There is nothing in the article here about how snot is needed to help differiate smells. Yes, mucus in intlligently designed. Artificial noses have an manmade mucus to it. I tellya. All my stays at Holiday Express really paying off.
[I love those old commericials. See on youtube.]