
Scientists have provided the most detailed account yet of the earliest stages of DNA replication, an essential process for all life to grow and reproduce.
For the first time, scientists have directly observed the very moment DNA begins to unravel, a critical molecular event that underpins its role as the carrier of genetic information. In a groundbreaking study published in Nature, researchers from King Abdullah University of Science and Technology (KAUST) have captured the initial steps of DNA replication, offering new insight into how cells accurately duplicate their genetic material, a process essential for life, growth, and reproduction.
Using advanced cryo-electron microscopy combined with deep learning techniques, the teams led by KAUST Assistant Professor Alfredo De Biasio and Professor Samir Hamdan closely examined how the helicase enzyme, Simian Virus 40 Large Tumor Antigen, interacts with DNA. Their work reveals 15 distinct atomic-level states that detail how the helicase initiates and drives the unwinding of the DNA double helix. This achievement marks a major breakthrough not only in understanding helicase function but also in visualizing enzyme dynamics at atomic resolution—an unprecedented step forward in molecular biology.
Understanding Helicase and Its Role
While scientists have long known the importance of helicase in DNA replication, “they did not know how DNA, helicases, and ATP work together in a coordinated cycle to drive DNA unwinding,” De Biasio said.
When Watson and Crick reported the double helix in 1953, they gave the scientific community a breakthrough understanding of how genetic information is stored and copied. For DNA to replicate, the helix must first unwind and break the DNA from a double strand into two single strands.
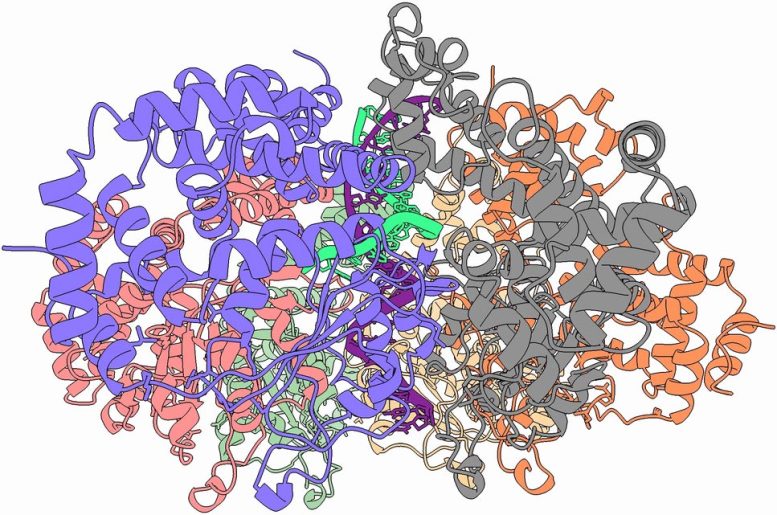
Upon binding, helicases melt the DNA, breaking the chemical bonds holding the double helix together. They then pull the two strands apart, allowing other enzymes to complete the replication. Without this first step, no DNA can be replicated. In this way, helicases are machines or, because of their size, nanomachines.
ATP: The Fuel Driving the Unwinding
If helicases are nanomachines, then ‘ATP’, or adenosine trisphosphate, is the fuel. Much like how burning gas drives the pistons of a car engine, burning ATP, the same fuel used to flex your muscles, causes the six pistons of a helicase to unwind DNA. The study found that as ATP is consumed, it reduces physical constraints that allow the helicase to proceed along the DNA, unwinding more and more of the double strand. Thus, ATP consumption acts a switch that increases the amount of entropy – or disorder – in the system, freeing the helicase to move along the DNA.
“The helicase uses ATP not to pry DNA apart in one motion, but to cycle through conformational changes that progressively destabilize and separate the strands. ATP burning, or hydrolysis, functions like the spring in a mouse trap, snapping the helicase forward and pulling the DNA strands apart,” said De Biasio.
Among the many discoveries made by the KAUST scientists was that two helicases melt the DNA at two sites at the same time to initiate the unwinding. The chemistry of DNA is such that nanomachines move along a single DNA strand in one direction only. By binding at two sites simultaneously, the helicases coordinate so that the winding can happen in both directions with an energy efficiency unique to natural nanomachines.
That efficiency, explains De Biasio, makes the study of DNA replication more than an attempt to answer the most fundamental scientific questions about life, it also makes helicase models for the design of new nanotechnology.
“From a design perspective, helicases exemplify energy-efficient mechanical systems. Engineered nanomachines using entropy switches could harness similar energy-efficient mechanisms to perform complex, force-driven tasks,” he said.
Reference: “Structural dynamics of DNA unwinding by a replicative helicase” by Taha Shahid, Ammar U. Danazumi, Muhammad Tehseen, Lubna Alhudhali, Alice R. Clark, Christos G. Savva, Samir M. Hamdan and Alfredo De Biasio, 19 March 2025, Nature.
DOI: 10.1038/s41586-025-08766-w
Never miss a breakthrough: Join the SciTechDaily newsletter.
Follow us on Google and Google News.
3 Comments
IE, there is a God who designs DNA and energy efficient nano machines.
Similarly RNA strands from viruses msy cause Mutations in DNA when the process of DNA replication occurs during which DNA strands get seperated and virus RNA attaches to it. Is it so.
GOD is like DUH