Researchers investigating a widely used medication have uncovered an unexpected cellular process that may interfere with early brain development.
A medication widely used to control epilepsy, migraines, and bipolar disorder has long carried a serious warning for pregnant patients. Valproic acid is known to increase the risk of birth defects, but scientists have struggled to explain exactly how this damage occurs. New research offers a clearer answer by pointing to an unexpected biological process that interferes with early brain development.
In a study published in PLOS Biology, researchers led by Bill Keyes at the Institute of Genetics and Molecular and Cellular Biology in France found that valproic acid (VPA) can force critical cells in a developing embryo into a state called senescence. In this condition, cells stop dividing and functioning as they should. While senescence is often linked to aging or the body’s response to damage, its appearance during early development can disrupt the formation of essential structures.
That disruption may help explain the wide range of problems seen in children exposed to VPA before birth. For decades, doctors have documented thousands of such cases, including neural tube defects like spina bifida, facial differences, and heart abnormalities. Research has also shown that roughly one in three exposed children later experience cognitive challenges or are diagnosed with Autism Spectrum Disorder.
Tracking the Cellular Effects
To investigate what is happening at the cellular level, the team combined experiments in mice with studies using human organoids, which are small, three-dimensional models of developing tissue grown in the lab. These models allowed the researchers to observe early brain development in detail. They found that VPA specifically affects neuroepithelial cells, a population of stem cells that normally multiply and give rise to the brain and spinal cord. Instead of continuing to grow, many of these cells entered senescence, effectively reducing the pool of cells needed to build a healthy nervous system.
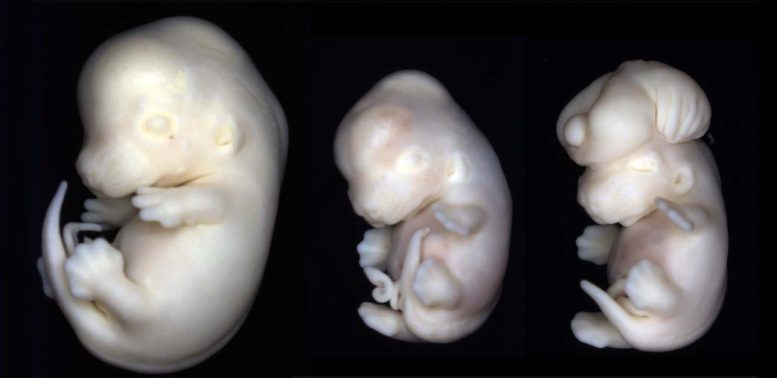
The researchers traced this effect to a molecule called p19Arf, which appears to act as a trigger for the senescence response. When mice were genetically modified to lack p19Arf, some of the most striking VPA-related effects disappeared. These animals did not develop microcephaly, a condition marked by reduced head size, and they avoided certain gene activity changes linked to Autism Spectrum Disorder. Even so, other abnormalities still occurred, suggesting that VPA disrupts development through multiple pathways.
Rethinking Senescence in Development
The findings highlight a lesser-known risk factor in embryonic development. Cellular senescence has traditionally been studied in the context of aging and cancer prevention, where it can serve as a protective mechanism. This study shows that the same process, when activated at the wrong time, may instead derail normal development.
“Overall, the discovery that atypical activation of senescence in the embryo can perturb development raises the intriguing possibility that it may also contribute to defects in developmental contexts beyond those we studied here.”
Muriel Rhinn, the study’s first author, emphasized the broader implications: “While cellular senescence has long been associated with aging and age-related disease, we now show that aberrant induction of senescence can also contribute to developmental defects. As valproic acid is strongly linked to cognitive defects and Autism Spectrum Disorder, this study now introduces an exciting link with senescence, supporting how additional studies are needed.”
The researchers have also published a follow-up paper in The FEBS Journal, expanding on these findings. In it, they argue that misregulated cellular senescence may play a broader role in developmental disorders, suggesting it could be an underrecognized driver of birth defects beyond those linked to valproic acid.
References:
“Aberrant induction of p19Arf-mediated cellular senescence contributes to neurodevelopmental defects” by Muriel Rhinn, Irene Zapata-Bodalo, Annabelle Klein, Jean-Luc Plassat, Tania Knauer-Meyer and William M. Keyes, 14 June 2022, PLOS Biology.
DOI: 10.1371/journal.pbio.3001664
“Cellular senescence and developmental defects” by Annabelle Klein, Muriel Rhinn and William M. Keyes, 1 March 2023, The FEBS Journal.
DOI: 10.1111/febs.16731
Never miss a breakthrough: Join the SciTechDaily newsletter.
Follow us on Google and Google News.
3 Comments
I dont know why scientists insist on using mice for experiments meant for humans the results will never be accurate (squeak)
The results will never be accuRAT! C:
Mostly it’s because they are a decent analog for humans that can have many generations very fast. They are only the first part of testing for human things. It’s not great, as is animal testing period, but it’s what we got right now.
Interesting study, even though it could not necessarily be applied to humans; however, it is only one drug under study, while they talk about other drugs that could be involved, without either naming them or at least suggesting which ones need to be studied.