Scientists have discovered that transplanted stem cell-derived brain cells may do far more than simply survive after a stroke.
A stem cell treatment helped mice recover from strokes by rebuilding damaged brain connections, restoring blood vessels, and improving movement, according to new research from the University of Zurich and the University of Southern California. The findings raise hopes that future therapies could one day repair stroke damage that is currently considered permanent.
Stroke remains one of the world’s leading causes of long-term disability. When blood flow to part of the brain is cut off, oxygen-starved cells die within minutes. Unlike skin or bone, the brain has only a limited ability to replace lost tissue, leaving many survivors with lifelong paralysis, speech problems, or memory loss.
Scientists have spent years searching for ways to help the brain rebuild itself. In the new study, researchers used neural progenitor cells, early-stage cells capable of developing into different types of brain tissue. The cells were created from induced pluripotent stem cells, which are adult human cells reprogrammed into a stem cell-like state.
The team transplanted these cells into the brains of mice one week after a stroke. That timing turned out to be critical. Earlier transplants survived poorly because the injured brain was still overwhelmed by inflammation and toxic chemical signals. Waiting several days allowed conditions to stabilize enough for the transplanted cells to take hold.
What happened next surprised the researchers.
New Neurons and Rebuilt Connections
Over five weeks, the transplanted cells survived, spread through nearby brain tissue, and matured mostly into functioning neurons. Many became GABAergic neurons, specialized inhibitory brain cells that help regulate neural activity and are heavily depleted after stroke. These cells are essential for balancing brain signaling, preventing excessive excitation, and coordinating movement.
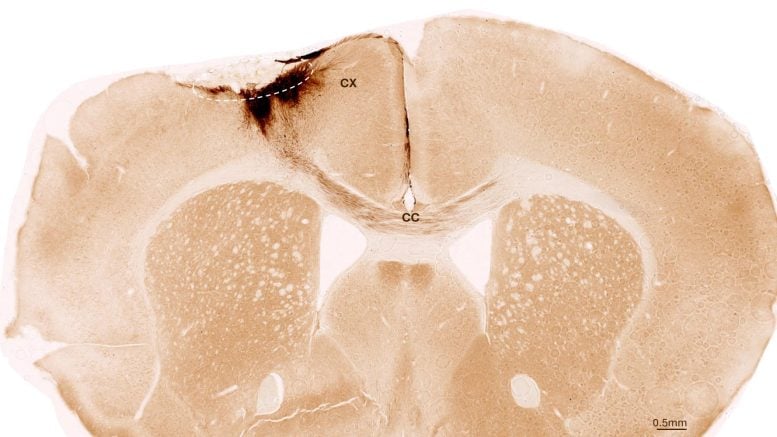
The grafted neurons did not simply exist alongside the damaged brain tissue. Evidence suggested they actively communicated with surrounding cells through molecular signaling systems linked to neural growth, synapse formation, and tissue repair. The researchers identified several major pathways involved in this cross-talk, including neurexin, neuregulin, NCAM, and SLIT signaling, all of which are associated with rebuilding neural networks and guiding axons to reconnect.
The stem cell treatment also appeared to trigger a broader healing response across the injured brain.
Mice receiving the transplants developed significantly more blood vessels near the stroke site, improving circulation in damaged tissue. The treatment also reduced inflammatory activity and strengthened the blood-brain barrier, the protective lining that normally prevents harmful substances in the bloodstream from leaking into the brain. Damage to this barrier is a major contributor to swelling and further injury after stroke.
Researchers additionally observed increased growth of nerve fibers around the damaged region. Some transplanted neurons extended long projections into areas linked to movement and sensory control, suggesting the new cells may have started integrating into existing brain circuits.
Improvements in Movement and Coordination
To measure whether these biological changes translated into real recovery, the scientists used AI-assisted motion tracking systems that analyzed how the mice walked. Stroke typically disrupts coordination, paw placement, and gait timing. Mice treated with stem cells gradually regained smoother movement and performed better on balance and fine-motor tasks than untreated animals.
The recovery was especially noticeable weeks after transplantation, indicating the therapy may support long-term repair rather than providing only short-lived benefits.
“Our findings show that neural stem cells not only form new neurons, but also induce other regeneration processes,” said Christian Tackenberg of the University of Zurich’s Institute for Regenerative Medicine.
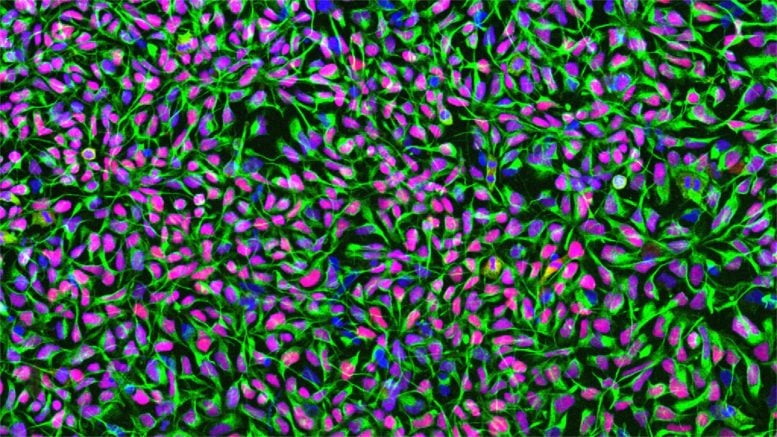
The study also addressed one of the biggest concerns surrounding stem cell therapies: safety. The neural progenitor cells were produced using animal-free methods designed for future clinical compatibility. Researchers are now developing built-in “safety switches” that could shut down transplanted cells if abnormal growth occurs.
Another major goal is making treatment less invasive. In the current experiments, cells were implanted directly into the brain. Scientists are now exploring whether stem cells could eventually be delivered through blood vessels instead, similar to minimally invasive stroke procedures already used in hospitals.
Several types of stem cell therapies have already reached early human trials for neurological diseases including Parkinson’s disease. Stroke may become one of the next major targets.
Important Questions Still Remain
Important hurdles remain. The experiments were performed in genetically modified mice whose immune systems would not reject human cells, and researchers have not yet proven that the transplanted neurons fully integrate into human-like brain networks over the long term.
“We need to minimize risks and simplify a potential application in humans,” Tackenberg said. “Stroke could be one of the next diseases for which a clinical trial becomes possible.”
Reference: “Neural xenografts contribute to long-term recovery in stroke via molecular graft-host crosstalk” by Rebecca Z. Weber, Beatriz Achón Buil, Nora H. Rentsch, Patrick Perron, Stefanie Halliday, Allison Bosworth, Mingzi Zhang, Kassandra Kisler, Chantal Bodenmann, Kathrin J. Zürcher, Daniela Uhr, Debora Meier, Siri L. Peter, Melanie Generali, Shuo Lin, Markus A. Rüegg, Roger M. Nitsch, Christian Tackenberg and Ruslan Rust, 16 September 2025, Nature Communications.
DOI: 10.1038/s41467-025-63725-3
Never miss a breakthrough: Join the SciTechDaily newsletter.
Follow us on Google and Google News.
9 Comments
So, from who, where would these “stem cells” come? Just curious..,
I had stroke 2018. I would like to have a chance to get arm & leg back l can move alitte bit. Iwould glad to any treament that will me get better.lm 70 yrs old l dont care.
I am currentlyust 46 years i don’t even have job to pay for physiotherapy session and my partner thinks this is the bedtime to as for a separation. So I need all that be done to give my hand back
Where can I get it im a person tha had a stroke about years ago do you believe it will work for me
Stem cell treatment has been studied and implemented for years. I know and have discussed this treatment with a doctor who’s famous father (Hockey Player) had it done. Also a famous Quarterback had it performed at the same clinic.
They are finally admitting this? My brain “told” me this a long time ago. It’s akin to road construction. Give it the materials and it will fix it.
My mother had a stroke 8 months ago. She is left with severe aphasia, but she has her memory. Just can’t get the words out. This is after she had covid
My husband has a TBI which is similar in ways to a stroke. I wonder if they have thought about this application to people that have brain damage due to accidents?
Let’s get moving on with the stem cell therapy. Why do we have to wait? And It shouldn’t be a expensive procedure.
Stem cell has been proven to heal many sport injuries. Knees, shoulders and spinal injuries. And to my surprise dental. So why wouldn’t it work for Strokes?
I would choose stem cell before doing a surgery. It’s less intrusive. I get it. Some times surgery is the only option.
Now science wants to install a chip in the brain for Stroke victims.they say it’s a faster recovery.. I question it.
I might choose the chip. If I didn’t have to suffer through years of rehabilitation.
I believe in stem cell therapy and it’s been a long time coming! I’m hoping it gets going!!
.
.