Specialized dye, delivered along with a vaccine, could enable “on-patient” storage of vaccination history.
Every year, a lack of vaccination leads to about 1.5 million preventable deaths, primarily in developing nations. One factor that makes vaccination campaigns in those nations more difficult is that there is little infrastructure for storing medical records, so there’s often no easy way to determine who needs a particular vaccine.
MIT researchers have now developed a novel way to record a patient’s vaccination history: storing the data in a pattern of dye, invisible to the naked eye, that is delivered under the skin at the same time as the vaccine.
“In areas where paper vaccination cards are often lost or do not exist at all, and electronic databases are unheard of, this technology could enable the rapid and anonymous detection of patient vaccination history to ensure that every child is vaccinated,” says Kevin McHugh, a former MIT postdoc who is now an assistant professor of bioengineering at Rice University.
The researchers showed that their new dye, which consists of nanocrystals called quantum dots, can remain for at least five years under the skin, where it emits near-infrared light that can be detected by a specially equipped smartphone.
McHugh and former visiting scientist Lihong Jing are the lead authors of the study, which was published on December 18, 2019, in Science Translational Medicine. Ana Jaklenec, a research scientist at MIT’s Koch Institute for Integrative Cancer Research, and Robert Langer, the David H. Koch Institute Professor at MIT, are the senior authors of the paper.
An invisible record
Several years ago, the MIT team set out to devise a method for recording vaccination information in a way that doesn’t require a centralized database or other infrastructure. Many vaccines, such as the vaccine for measles, mumps, and rubella (MMR), require multiple doses spaced out at certain intervals; without accurate records, children may not receive all of the necessary doses.
“In order to be protected against most pathogens, one needs multiple vaccinations,” Jaklenec says. “In some areas in the developing world, it can be very challenging to do this, as there is a lack of data about who has been vaccinated and whether they need additional shots or not.”
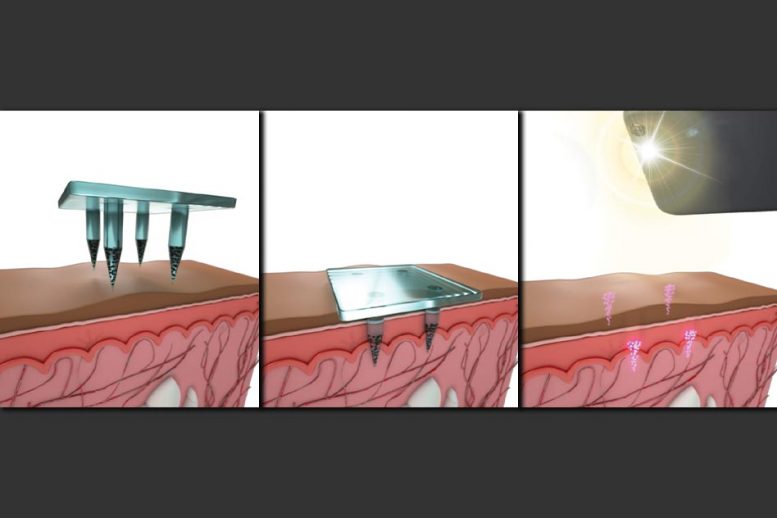
To create an “on-patient,” decentralized medical record, the researchers developed a new type of copper-based quantum dots, which emit light in the near-infrared spectrum. The dots are only about 4 nanometers in diameter, but they are encapsulated in biocompatible microparticles that form spheres about 20 microns in diameter. This encapsulation allows the dye to remain in place, under the skin, after being injected.
The researchers designed their dye to be delivered by a microneedle patch rather than a traditional syringe and needle. Such patches are now being developed to deliver vaccines for measles, rubella, and other diseases, and the researchers showed that their dye could be easily incorporated into these patches.
The microneedles used in this study are made from a mixture of dissolvable sugar and a polymer called PVA, as well as the quantum-dot dye and the vaccine. When the patch is applied to the skin, the microneedles, which are 1.5 millimeters long, partially dissolve, releasing their payload within about two minutes.
By selectively loading microparticles into microneedles, the patches deliver a pattern in the skin that is invisible to the naked eye but can be scanned with a smartphone that has the infrared filter removed. The patch can be customized to imprint different patterns that correspond to the type of vaccine delivered.
“It’s possible someday that this ‘invisible’ approach could create new possibilities for data storage, biosensing, and vaccine applications that could improve how medical care is provided, particularly in the developing world,” Langer says.
Effective immunization
Tests using human cadaver skin showed that the quantum-dot patterns could be detected by smartphone cameras after up to five years of simulated sun exposure.
The researchers also tested this vaccination strategy in rats, using microneedle patches that delivered the quantum dots along with a polio vaccine. They found that those rats generated an immune response similar to the response of rats that received a traditional injected polio vaccine.
“This study confirmed that incorporating the vaccine with the dye in the microneedle patches did not affect the efficacy of the vaccine or our ability to detect the dye,” Jaklenec says.
The researchers now plan to survey health care workers in developing nations in Africa to get input on the best way to implement this type of vaccination record keeping. They are also working on expanding the amount of data that can be encoded in a single pattern, allowing them to include information such as the date of vaccine administration and the lot number of the vaccine batch.
The researchers believe the quantum dots are safe to use in this way because they are encapsulated in a biocompatible polymer, but they plan to do further safety studies before testing them in patients.
“Storage, access, and control of medical records is an important topic with many possible approaches,” says Mark Prausnitz, chair of chemical and biomolecular engineering at Georgia Tech, who was not involved in the research. “This study presents a novel approach where the medical record is stored and controlled by the patient within the patient’s skin in a minimally invasive and elegant way.”
Reference: “Biocompatible near-infrared quantum dots delivered to the skin by microneedle patches record vaccination” by Kevin J. McHugh, Lihong Jing, Sean Y. Severt, Mache Cruz, Morteza Sarmadi, Hapuarachchige Surangi N. Jayawardena, Collin F. Perkinson, Fridrik Larusson, Sviatlana Rose, Stephanie Tomasic, Tyler Graf, Stephany Y. Tzeng, James L. Sugarman, Daniel Vlasic, Matthew Peters, Nels Peterson, Lowell Wood, Wen Tang, Jihyeon Yeom, Joe Collins, Philip A. Welkhoff, Ari Karchin, Megan Tse, Mingyuan Gao, Moungi G. Bawendi, Robert Langer and Ana Jaklenec, 18 December 2019, Science Translational Medicine.
DOI: 10.1126/scitranslmed.aay7162
The research was funded by the Bill and Melinda Gates Foundation and the Koch Institute Support (core) Grant from the National Cancer Institute. Other authors of the paper include Sean Severt, Mache Cruz, Morteza Sarmadi, Hapuarachchige Surangi Jayawardena, Collin Perkinson, Fridrik Larusson, Sviatlana Rose, Stephanie Tomasic, Tyler Graf, Stephany Tzeng, James Sugarman, Daniel Vlasic, Matthew Peters, Nels Peterson, Lowell Wood, Wen Tang, Jihyeon Yeom, Joe Collins, Philip Welkhoff, Ari Karchin, Megan Tse, Mingyuan Gao, and Moungi Bawendi.
Never miss a breakthrough: Join the SciTechDaily newsletter.
Follow us on Google and Google News.
2 Comments
Yes USA!
Sounds “Orwellian”. I do not like this one little bit.