An experiment at the State University of Campinas found that FGF19, a substance produced in the intestine, acts on specific brain regions to trigger energy burning and heat production, paving the way for the development of new drugs.
Recent research on mice has uncovered how a hormone produced in the intestine communicates with the brain to help regulate the body’s use of energy. The hormone, known as FGF19 (fibroblast growth factor 19), activates processes that encourage the body to expend more energy, burn stored fat, and support healthier weight and blood sugar levels in obese animals.
Scientists linked these effects to FGF19’s activity in the hypothalamus, a region of the brain responsible for coordinating signals from both the body and the environment to control energy metabolism. The study showed that when FGF19 interacts with the hypothalamus, it increases the activity of thermogenic adipocytes (i.e., fat cells that burn energy to produce heat).
This finding could guide the creation of new treatments for obesity, diabetes, and other metabolic conditions by using compounds that imitate the actions of substances naturally produced by the body.
Such an approach resembles advanced diabetes medications already in use for weight management. One well-known example is Ozempic, which contains the active ingredient semaglutide. This compound activates receptors that mimic the hormone GLP-1, sending satiety signals to the brain and helping control appetite.
Thermogenesis and Inflammation
The study also found that FGF19 reduced inflammation in body tissues and improved tolerance to cold. However, these positive effects disappeared when the sympathetic nervous system was experimentally suppressed. Researchers observed that exposure to cold increased the number of FGF19 receptors in the hypothalamus, a brain region central to temperature regulation. This suggests that FGF19 may help the body adapt by balancing energy use and maintaining thermal stability.
“FGF19 had already been linked to a reduction in food intake. Our work broke new ground by showing that it also plays an important role by acting on the hypothalamus and stimulating an increase in energy expenditure in white and brown adipose tissue. In other words, in addition to controlling appetite, it stimulates thermogenesis. So, in terms of therapy associated with obesity, it’d make a lot of sense,” explains Professor Helena Cristina de Lima Barbosa, from the Obesity and Comorbidities Research Center (OCRC) at the State University of Campinas (UNICAMP).
The OCRC is a Research, Innovation, and Dissemination Center (RIDC) of FAPESP, which also supported the study through grants awarded to doctoral student Lucas Zangerolamo, first author of the work, under Barbosa’s guidance.
The article describing these findings has been published in the American Journal of Physiology – Endocrinology and Metabolism and was highlighted by the journal as a Top Article in May.
A Worldwide Health Crisis
The World Atlas of Obesity 2025 points out that based on current trends, the world will not achieve this year’s targets for preventing and controlling chronic non-communicable diseases. These goals include stopping the increase in diabetes and obesity, as well as reducing premature mortality from cardiovascular, chronic respiratory, and cancer diseases by 25%, using 2010 data as a baseline.
According to the Atlas, over 1 billion people worldwide live with obesity. Projections indicate that this number could exceed 1.5 billion by 2030 if effective measures are not implemented. Obesity is linked to 1.6 million premature deaths per year caused by non-communicable diseases.
In Brazil, approximately 31% of people are obese, and between 40% and 50% of adults do not exercise at the recommended frequency or intensity.
Step-by-step research
FGF19, which is involved in the control of energy metabolism, is mainly produced in the small intestine. In the liver, it regulates the production of bile acids, as well as the synthesis of glucose and fats. While its primary functions in the liver have been well-documented in scientific literature, its signaling in the brain has received limited analysis.
“In the lab, we work with bile acids, which are also the subject of my master’s degree, and they regulate the release of FGF-19. Our initial studies led us down this path,” Zangerolamo told Agência FAPESP.
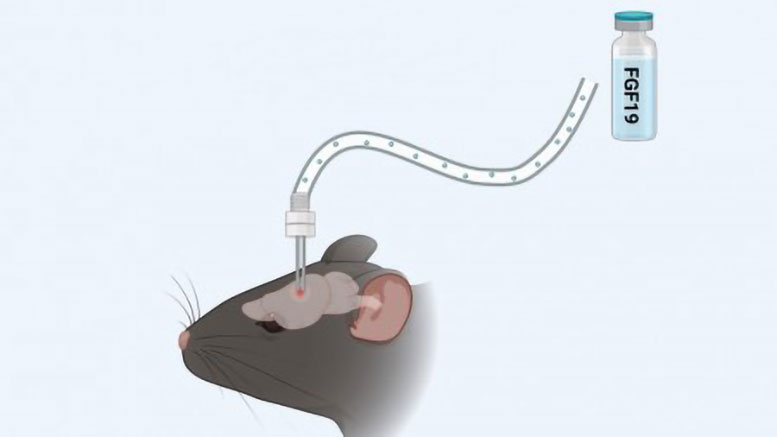
At eight weeks of age, the animals were randomly divided into two groups: one with a regular diet (control) and one with a high-fat diet. The hormone was administered directly into the brains of the obese mice. The animals were housed in an environment controlled for temperature, lighting, and water supply.
In the article, the scientists point out that central FGF19 signaling improved energy homeostasis by increasing sympathetic nervous system activity and stimulating adipose tissue thermogenesis, causing the tissue to burn more energy in the form of heat.
“The brain plays an extremely important role in controlling the body’s adiposity. At the same time as it receives information from peripheral tissues, it triggers commands. These commands, apparently using the sympathetic nervous system, seem to be an interesting way of thinking about energy expenditure,” adds Barbosa.
The authors compiled and analyzed public scRNA-seq data from different papers on the hypothalamus. This technique allows the RNA of individual cells to be sequenced. The authors analyzed the transcription of more than 50,000 single cells to identify hypothalamic cell types that express FGF19 receptors.
The researchers explain that a key issue now is understanding how to stimulate the body to produce more FGF19. The group continues to work to understand how the pathways involved in eating behavior are linked to this process.
“We want to broaden this understanding. We’re studying the hypothalamus to evaluate the inflammation commonly observed when a high-fat diet is administered and whether FGF19 plays a role in this area,” says Zangerolamo, who did part of the work during an internship at the Joslin Diabetes Center at Harvard Medical School with Professor Yu-Hua Tseng, who is also an author of the article.
Reference: “Central FGF19 signaling enhances energy homeostasis and adipose tissue thermogenesis through sympathetic activation in obese mice” by Lucas Zangerolamo, Marina Carvalho, Carina Solon, Davi Sidarta-Oliveira, Gabriela M. Soares, Carine Marmentini, Antonio C. Boschero, Yu-Hua Tseng, Licio A. Velloso and Helena C. L. Barbosa, 1 April 2025, American Journal of Physiology-Endocrinology and Metabolism.
DOI: 10.1152/ajpendo.00488.2024
Funding: São Paulo Research Foundation
Never miss a breakthrough: Join the SciTechDaily newsletter.
Follow us on Google and Google News.
1 Comment
Comet Haley Kaitlin Smith Brothere Back drop Pictures People Photography Subjects Photograph Photographer Album Dark room developer dye Thai Cuisine motion film sets Network Abc Campbell Building pop Articulations article confederate railroad retirement Pension plans wedding announcement singer funeral signed permission slip arrest records dress barn Yard animal Planet Earth girls are easy bake oven appliances annual Maytag whirlpool wascher Patent theft digital Millianiaum and act smart phone East t street apartments Armed Forces Removal Forcibly removed Webster Publist Alley hooker hooker initial t tablet pillage village I ik