
Scientists have developed an innovative technique that combines expansion microscopy with mass spectrometry imaging to visualize hundreds of biomolecules in intact tissues at single-cell resolution.
This breakthrough could transform our understanding of biological processes like aging and disease by revealing the molecular layout within tissues. With no need for specialized equipment, the method is easy to adopt, opening new doors for labs around the world.
Challenges in Visualizing Molecules
For biologists, seeing is essential to understanding, but it’s not always easy.
A major challenge is visualizing all the different molecules within an intact tissue sample, all the way down to individual cells. Being able to pinpoint the location of hundreds or even thousands of biomolecules, such as lipids, proteins, and metabolites, in their natural environment helps researchers study how these molecules function and interact. Yet, existing tools fall short of delivering this comprehensive view.
Standard imaging methods, like various forms of microscopy, allow scientists to look inside cells, but they’re limited to tracking only a few specific molecules at once. Some biomolecules, especially certain lipids, are difficult to detect with these techniques. On the other hand, traditional mass spectrometry can detect a broad range of molecules, but it requires breaking down the tissue, making it impossible to see how those molecules are spatially arranged.
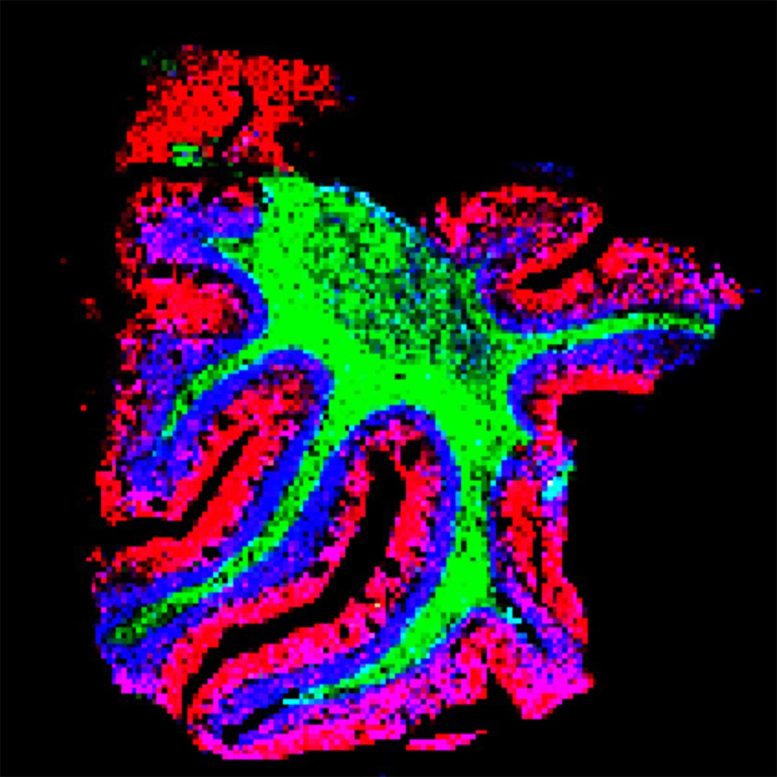
Introducing Mass Spectrometry Imaging
Mass spectrometry imaging offers a partial solution. It allows researchers to view many different biomolecules at once within intact tissue samples. However, its resolution isn’t high enough to distinguish molecular details at the single-cell level.
This was the problem facing Meng Wang, a Senior Group Leader at Janelia. Her team studies the biological mechanisms that drive aging and longevity, and they needed a way to observe a wide array of biomolecules in whole tissues to understand how these components change over time.
“Knowing at each specific location what molecules are there and what is in the neighboring cells is very important for any kind of biological question,” Wang explains.
Expansion Microscopy as a Game Changer
Luckily, Wang’s lab is down the hall from Janelia Principal Scientist Paul Tillberg. Tillberg co-invented a technique called expansion microscopy as a graduate student at MIT. The method uses a swellable hydrogel material to expand samples uniformly in all directions to a point where fine details, like sub-organelle structure, can be detected with a conventional microscope.
Now a decade old, the expansion process is being applied to other methods outside traditional microscopy. Wang, Tillberg, and their collaborators at Janelia and the University of Wisconsin-Madison wanted to see if they could use expansion to overcome mass spectrometry imaging’s spatial resolution problem.
Seeing Molecules at Single-Cell Resolution
The result is a new method that expands tissue samples gradually without having to degrade them at the molecular level, as happens in the original expansion process. By expanding the intact samples in all directions, researchers can use mass spectrometry imaging to simultaneously detect hundreds of molecules at the single-cell level in their native locations.
“This lets you have an untargeted look in the molecular space, and we are trying to bring it closer to what microscopy can do in terms of spatial resolution,” Tillberg says.
Mapping Molecular Signatures in the Brain
The team used the new technique to delineate the specific spatial patterns of small molecules in different layers of the cerebellum. They found that these molecules – including lipids, peptides, proteins, metabolites, and glycans – are not uniformly distributed, as previously thought. Moreover, they found that each specific layer of the cerebellum has its own signature of lipids, metabolites, and proteins.
The team was also able to detect biomolecules in kidney, pancreas, and tumor tissues, demonstrating that the method can be adapted for many different tissue types. In tumor tissues, they were able to visualize large variations in biomolecules, which could be useful for understanding the molecular mechanisms of tumors and potentially aid in drug development.
“When you can see these biomolecules, then you can start to understand why they have such patterns and how that is related to function,” says Wang. She believes the new technology will allow researchers to track these patterns during development, aging, and disease to understand how different molecules contribute to these processes.
Broad Accessibility and Future Potential
Because the new method doesn’t require adding hardware to an existing mass spec imaging system, and the expansion technique is relatively easy to learn, the team hopes it will be used by many labs around the world. They also hope the new technique will make mass spec imaging a more useful tool for biologists and have laid out a detailed description of the new method and a roadmap for adapting it to other tissue types.
“We wanted to develop something that did not require specialized instruments or procedures, but can be broadly adopted,” Wang says.
Reference: “TEMI: tissue-expansion mass-spectrometry imaging” by Hua Zhang, Lang Ding, Amy Hu, Xudong Shi, Penghsuan Huang, Haiyan Lu, Paul W. Tillberg, Meng C. Wang and Lingjun Li, 22 April 2025, Nature Methods.
DOI: 10.1038/s41592-025-02664-9
Never miss a breakthrough: Join the SciTechDaily newsletter.
Follow us on Google and Google News.
1 Comment
Terrific entertainment