
Technology can be harnessed to design novel and enhanced molecules that address challenges in biotechnology or medicine.
Australian researchers, including those at the Charles Perkins Centre at the University of Sydney, have created a groundbreaking system that uses what they describe as “biological artificial intelligence” to develop and refine molecules with enhanced or entirely new functions inside mammalian cells. According to the team, this innovation offers scientists a powerful way to create more precise research tools and targeted gene therapies.
The system, called PROTEUS (PROTein Evolution Using Selection), is built on a method known as directed evolution—a laboratory process that simulates how natural evolution works. Unlike natural evolution, which can take years or longer, PROTEUS speeds up this process dramatically, enabling the development of novel molecules in just a matter of weeks.
This advancement holds promising potential for improving medical treatments. One example is using PROTEUS to enhance the performance of gene-editing technologies such as CRISPR, increasing their precision and effectiveness in therapeutic applications.
“This means PROTEUS can be used to generate new molecules that are highly tuned to function in our bodies, and we can use it to make new medicine that would be otherwise difficult or impossible to make with current technologies.” says co-senior author Professor Greg Neely, Head of the Dr. John and Anne Chong Lab for Functional Genomics at the University of Sydney.
“What is new about our work is that directed evolution primarily works in bacterial cells, whereas PROTEUS can evolve molecules in mammal cells.”
Biological AI That Solves Molecular Problems
PROTEUS can be given a problem with uncertain solution like when a user feeds in prompts for an artificial intelligence platform. For example the problem can be how to efficiently turn off a human disease gene inside our body.
PROTEUS then uses directed evolution to explore millions of possible sequences that have yet to exist naturally and finds molecules with properties that are highly adapted to solve the problem. This means PROTEUS can help find a solution that would normally take a human researcher years to solve, if at all.

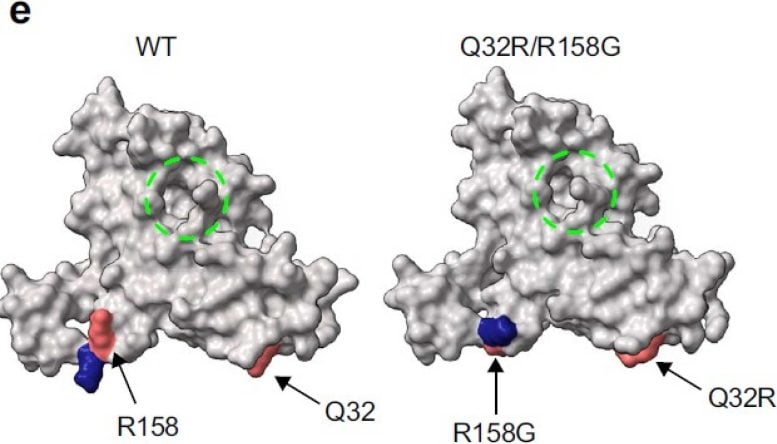
The researchers reported they used PROTEUS to develop improved versions of proteins that can be more easily regulated by drugs, and nanobodies (mini versions of antibodies) that can detect DNA damage, an important process that drives cancer. However, they said PROTEUS isn’t limited to this and can be used to enhance the function of most proteins and molecules.
The findings were reported in Nature Communications, with the research performed at the Charles Perkins Centre, the University of Sydney with collaborators from the Centenary Institute.
Unlocking molecular machine learning
The original development of directed evolution, performed first in bacteria, was recognized by the 2018 Noble Prize in Chemistry.
“The invention of directed evolution changed the trajectory of biochemistry. Now, with PROTEUS, we can program a mammalian cell with a genetic problem we aren’t sure how to solve. Letting our system run continuously means we can check in regularly to understand just how the system is solving our genetic challenge,” said lead researcher Dr. Christopher Denes from the Charles Perkins Center and School of Life and Environmental Sciences
The biggest challenge Dr. Denes and the team faced was how to make sure the mammalian cell could withstand the multiple cycles of evolution and mutations and remain stable, without the system “cheating” and coming up with a trivial solution that doesn’t answer the intended question.
They found the key was using chimeric virus-like particles, a design consisting of taking the outside shell of one virus and combining it with the genes of another virus, which blocked the system from cheating.
The design used parts of two significantly different virus families creating the best of both worlds. The resulting system allowed the cells to process many different possible solutions in parallel, with improved solutions winning and becoming more dominant while incorrect solutions instead disappear.
“PROTEUS is stable, robust, and has been validated by independent labs. We welcome other labs to adopt this technique. By applying PROTEUS, we hope to empower the development of a new generation of enzymes, molecular tools, and therapeutics,” Dr. Denes said.
“We made this system open source for the research community, and we are excited to see what people use it for. Our goals will be to enhance gene-editing technologies, or to fine-tune mRNA medicines for more potent and specific effects,” Professor Neely said.
Reference: “A chimeric viral platform for directed evolution in mammalian cells” by Alexander J. Cole, Christopher E. Denes, Cesar L. Moreno, Lise Hunault, Thomas Dobson, Daniel Hesselson and G. Gregory Neely, 7 May 2025, Nature Communications.
DOI: 10.1038/s41467-025-59438-2
Declaration: Alexandar Cole, Christopher Denes, Daniel Hesselson and Greg Neely have filed a provisional patent application on this technology.
Never miss a breakthrough: Join the SciTechDaily newsletter.
1 Comment
Could, but won’t.