
Researchers have found a potential game-changing way to help people lose weight and control blood sugar without the unpleasant side effects of today’s popular drugs like Ozempic.
- Current drug challenges: Popular weight loss drugs like GLP-1s (Ozempic, Wegovy, Mounjaro, Zepbound, etc.) often cause nausea and vomiting, leading most people to stop treatment within a year. Scientists are working on safer, more tolerable options.
- A surprising brain connection: Researchers discovered that brain support cells naturally produce a molecule that helps reduce appetite. They created a new version of this molecule, which helped lab animals lose weight and improve blood sugar control—without making them sick.
- A promising new path: This approach could lead to a new generation of weight loss treatments that deliver results without the unpleasant side effects, like finding a faster, easier route to the finish line.
Limitations of Current Weight Loss Drugs
Many current medications for weight loss and diabetes fail to help patients keep the weight off in the long run. Popular GLP-1 drugs work by acting on brain neurons that regulate appetite, but they often cause uncomfortable side effects. For around 70% of people, nausea and vomiting become severe enough to make them quit the treatment within a year.
Robert Doyle, a chemistry professor at Syracuse University, is leading a team of experts from multiple fields in search of a better approach. Their goal is to target a different part of the brain that could support weight loss without triggering gastrointestinal discomfort.
In most brain research, neurons are the primary focus. GLP-1 drugs, for instance, act on neurons in the hindbrain that help control hunger. However, scientists are beginning to explore the role of other brain cells known as “support” cells, including glia and astrocytes, as possible allies in reducing appetite.
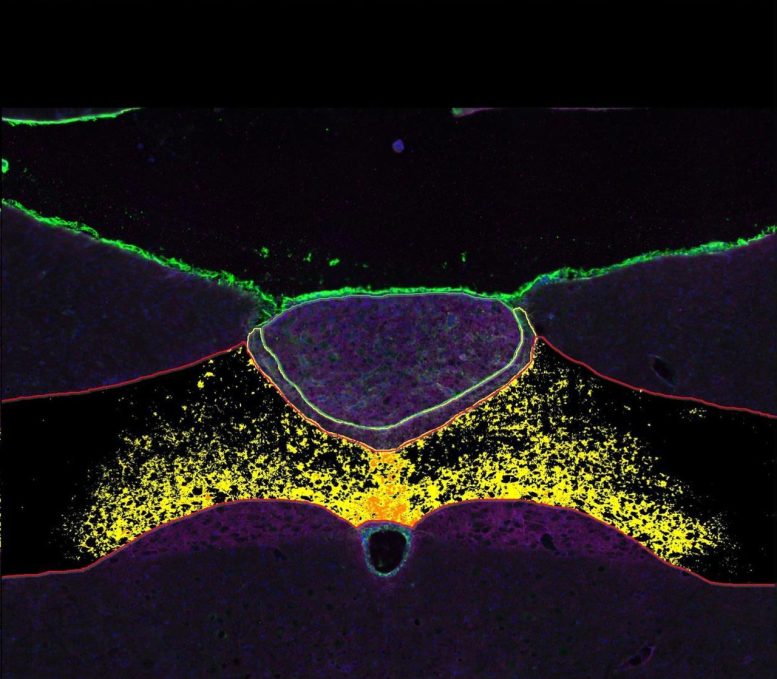
Support Cells and Hunger Reduction
Joint research has shown that these support cells can influence feelings of hunger, although this area has not been widely explored.
“We wanted to know whether support cells might produce new peptides or new signaling molecules that might be critical in body weight reduction,” says Doyle, a medicinal chemist and the Jack and Laura H. Milton Professor of Chemistry in the College of Arts and Sciences at Syracuse University. Doyle is also a professor of pharmacology and medicine at SUNY Upstate Medical University.
Working with colleagues at the University of Pennsylvania and the University of Kentucky, Doyle helped identify a chemical produced by astrocytes that could open the door to future appetite-control drugs with fewer side effects. Their findings were published in Science Translational Medicine.

How It Works: Brain “Light Bulbs” and Support Wiring
Think of each brain neuron as a light bulb and support cells as the components that allow the light bulb to brighten, including the wiring, switch, and filament.
“All of those supporting parts beyond the light bulb play a role in making the light shine,” says Doyle.
The research team discovered that some support cells in the hindbrain naturally produce a molecule named octadecaneuropeptide (ODN), which suppresses appetite. In lab tests, injecting ODN directly into rats’ brains made them lose weight and improved how they processed glucose.
However, injecting directly into the brain isn’t a practical treatment for people, so researchers created a new version of the molecule named tridecaneuropeptide (TDN). This molecule version could be given to human patients through regular injections akin to today’s Ozempic or Zepbound. When tested in obese mice and musk shrews, TDN helped the animals lose weight and respond better to insulin without causing nausea or vomiting.
Marathon Shortcut: Targeting Downstream Pathways
One goal of the research team is to produce weight loss without aiming new therapeutic molecules at neurons. The new TDN molecule bypasses neurons, taking a shortcut to directly target neurons’ downstream support cells, which researchers found also produce appetite suppression. TDN cuts short the “marathon” of chemical reactions and negative side-effects caused by GLP-1 drugs.
“Instead of running a marathon from the very beginning like current drugs do, our targeting downstream pathways in support cells is like starting the race halfway through, reducing the unpleasant side effects many people experience,” says Doyle. “If we could hit that downstream process directly, then potentially we wouldn’t have to use GLP-1 drugs with their side effects. Or we could reduce their dose, improving the toleration of these drugs. We could trigger weight loss signals that happen later in the pathway more directly.”
From Lab Breakthrough to Real-World Treatment
A new company called CoronationBio has been launched to turn this discovery into a real-world treatment. The company has licensed intellectual property related to ODN derivatives for the treatment of obesity and cardio-metabolic disease from Syracuse University and the University of Pennsylvania, with a focus on translating candidates into the clinic. They’re now teaming up with other companies to develop this treatment and aim to start human trials in 2026 or 2027.
Reference: “Hindbrain octadecaneuropeptide gliotransmission as a therapeutic target for energy balance control without nausea or emesis” by Caroline E. Geisler, Kylie S. Chichura, Oleksandr Orativskyi, Jiayin Hu, Drew L. Belser, Caitlyn M. Pelletier, Tito Borner, Caitlin Baumer-Harrison, Bart C. De Jonghe, Richard C. Crist, Benjamin C. Reiner, Robert P. Doyle and Matthew R. Hayes, 23 July 2025, Science Translational Medicine.
DOI: 10.1126/scitranslmed.adu6764
Never miss a breakthrough: Join the SciTechDaily newsletter.
Follow us on Google and Google News.
2 Comments
Need a drug that both reduces appetite and makes the patient antsy so that exercise happens almost automatically to relieve the hyper energetic feelings.
{^_^}
Yes we need a drug both reduce happy and that doesn’t make people 😷 sicy