
Researchers at Empa have taken a significant step toward practical quantum computing by creating and manipulating Heisenberg spin chains with nanographene molecules.
The smallest unit of information in a computer is the bit—either on or off, 1 or 0. Modern computing power is built entirely on the combination and interconnection of countless ones and zeros. Quantum computers, however, have their own version of the bit: the qubit. Like a classical bit, a qubit has two basic states. The key difference is that quantum effects enable superposition, allowing a qubit to exist as both 0 and 1 simultaneously, in varying proportions. This means a qubit can theoretically represent an infinite number of states.
This ambiguity is what gives quantum computers their theoretical “superpowers.” In principle, quantum computers can solve problems in mere fractions of a second—problems that would take today’s most powerful supercomputers an impractical amount of time. However, quantum computing is still in its early stages. One of the biggest challenges is connecting qubits, as a single qubit alone cannot function as a computer.
One way to represent the 0 and 1 of a qubit is through the alignment of electron spin, a fundamental quantum mechanical property of electrons and other particles. Simply put, spin can be thought of as a kind of torque, pointing either “up” (1) or “down” (0). When two or more spins are quantum-mechanically entangled, they influence each other’s states—changing the orientation of one affects all the others. This makes spin interactions a promising way to enable qubits to “communicate.”
However, as with much of quantum physics, this “language”—the interaction between spins—is extraordinarily complex. While it can be described mathematically, solving the equations exactly is nearly impossible, even for relatively simple chains of just a few spins. Not exactly ideal conditions for turning theory into reality…
A model becomes reality
Researchers at Empa’s nanotech@surfaces laboratory have now developed a method that allows many spins to “talk” to each other in a controlled manner – and that also enables the researchers to “listen” to them, i.e. to understand their interactions. Together with scientists from the International Iberian Nanotechnology Laboratory and the Technical University of Dresden, they were able to precisely create an archetypal chain of electron spins and measure its properties in detail. Their results have now been published in the renowned journal Nature Nanotechnology.
The theory behind the chain is familiar to all physics students: Take a linear chain of spins in which each spin interacts strongly with one of its neighbors and weakly with the other. This so-called one-dimensional alternating Heisenberg model was described almost 100 years ago by physicist and later Nobel Prize laureate Werner Heisenberg, one of the founders of quantum mechanics. Although there are materials in nature that contain such spin chains, it has not yet been possible to deliberately incorporate the chains into a material. “Real materials are always much more complex than a theoretical model,” explains Roman Fasel, head of Empa’s nanotech@surfaces laboratory and co-author of the study.
A “goblet” made of carbon
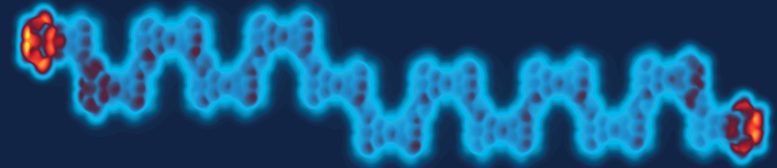
To create such an artificial quantum material, the Empa researchers used tiny pieces of the two-dimensional carbon material graphene. The shape of these nanographene molecules influences their physical properties, in particular, their spin – a kind of nano-sized quantum Lego brick from which the scientists can assemble longer chains.
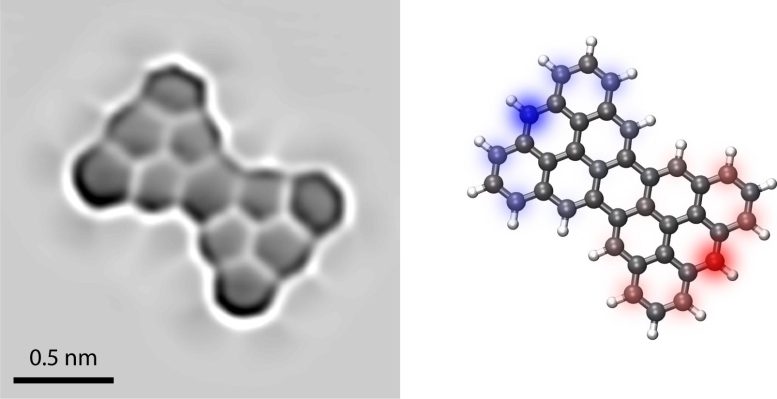
For their Heisenberg model, the researchers used the so-called Clar’s Goblet molecule. This special nanographene molecule consists of eleven carbon rings arranged in an hourglass-like shape. Due to this shape, there is an unpaired electron at each end – each with an associated spin. Although predicted by chemist Erich Clar as early as 1972, Clar’s Goblet was only produced in 2019 by Fasel’s team at the nanotech@surfaces laboratory.
The researchers have now linked the goblets on a gold surface to form chains. The two spins within a molecule are weakly linked, while the spins from molecule to molecule are strongly linked – a perfect realization of the alternating Heisenberg chain. The researchers were able to precisely manipulate the length of the chains, selectively switch individual spins on and off, and “flip” them from one state to another, allowing them to investigate the complex physics of this novel quantum material in great detail.
From theory to practice
Fasel is convinced that, just as the synthesis of Clar’s Goblet enabled the production of Heisenberg chains, this study will in turn open new doors in quantum research. “We have shown that theoretical models of quantum physics can be realized with nanographenes to test their predictions experimentally,” says the researcher. “Nanographenes with other spin configurations can be linked to form other types of chains or even more complex systems.” The Empa researchers are leading by example: In a second study, which is about to be published, they were able to recreate a different type of Heisenberg chain in which all spins are equally linked.
To be at the forefront of applied quantum physics, theoretical and experimental scientists from different disciplines need to work together. Chemists at Dresden University of Technology provided Empa researchers with the starting molecules for their synthesis of Clar’s Goblets. And researchers from the International Iberian Nanotechnology Laboratory in Portugal contributed their theoretical expertise to the project. The theory needed for such breakthroughs is not (just) what you find in physics textbooks, Fasel emphasizes, but a sophisticated transfer between the quantum physics model and the experimental measurements.
Reference: “Tunable topological phases in nanographene-based spin-1/2 alternating-exchange Heisenberg chains” by Chenxiao Zhao, Gonçalo Catarina, Jin-Jiang Zhang, João C. G. Henriques, Lin Yang, Ji Ma, Xinliang Feng, Oliver Gröning, Pascal Ruffieux, Joaquín Fernández-Rossier and Roman Fasel, 28 October 2024, Nature Nanotechnology.
DOI: 10.1038/s41565-024-01805-z
Never miss a breakthrough: Join the SciTechDaily newsletter.
Follow us on Google and Google News.