
The ocean’s smallest organisms could hold the biggest clues to Earth’s climate future.
The ocean’s tiniest engineers, calcifying plankton, play a vital yet often unnoticed role in regulating Earth’s climate by capturing and recycling carbon.
A new review published in Science by an international research team led by the Institute of Environmental Science and Technology at the Universitat Autònoma de Barcelona (ICTA-UAB) reveals that these organisms—coccolithophores, foraminifers, and pteropods—are oversimplified or excluded in many of the climate models used to forecast the planet’s future.
Overlooked impact on carbon cycling
By leaving out these plankton, current climate models may fail to account for crucial mechanisms in the global carbon cycle and underestimate the ocean’s ability to adapt to climate change. Calcifying plankton form delicate shells made of calcium carbonate (CaCO₃), which are central to the ocean’s carbon balance.
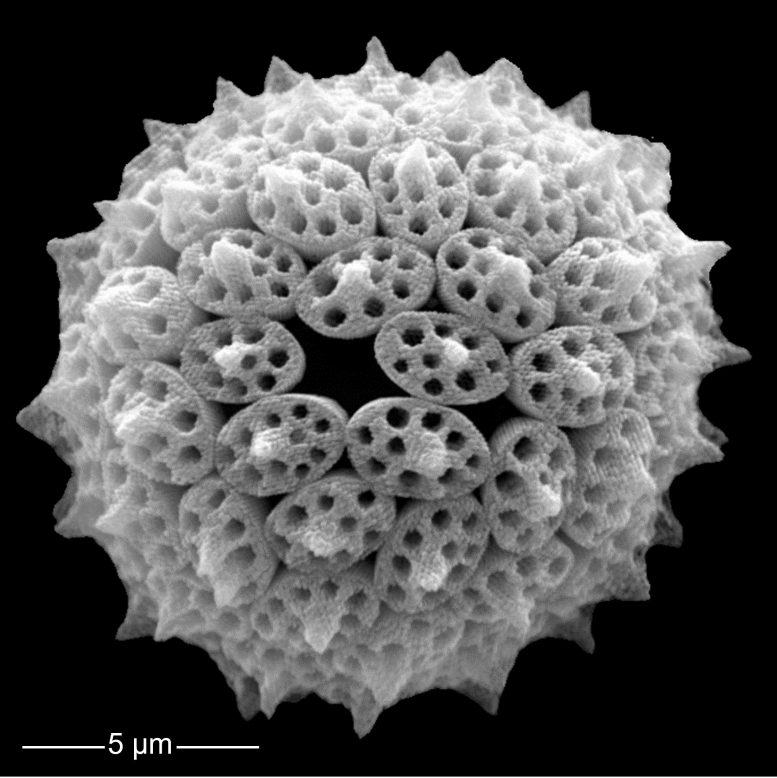
Through their life processes, they alter seawater chemistry and drive the movement of carbon from the atmosphere into the deep ocean. This natural “carbon pump” helps stabilize Earth’s climate and has long influenced both ocean chemistry and the fossil record.
“Plankton shells are tiny, but together they shape the chemistry of our oceans and the climate of our planet,” said Patrizia Ziveri, ICREA research professor at ICTA-UAB and lead author of the study. “By leaving them out of climate models, we risk overlooking fundamental processes that determine how the Earth system responds to climate change.”
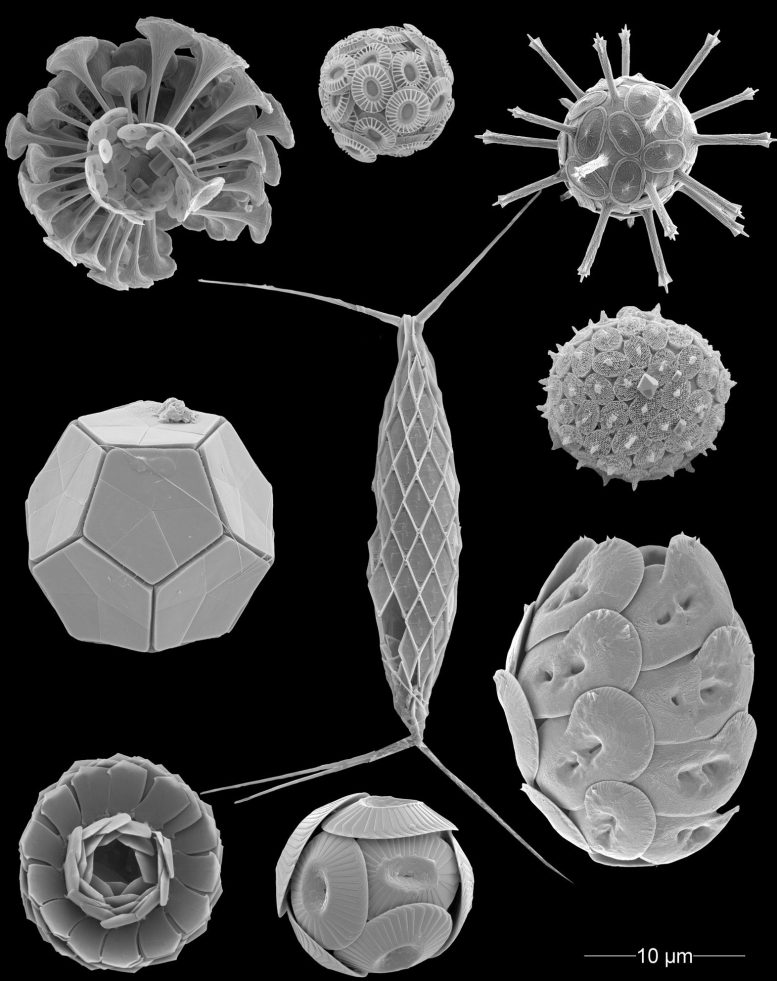
The researchers also found that much of the calcium carbonate produced by these organisms never settles on the ocean floor. Instead, a large portion dissolves in the upper layers of the ocean through a process known as “shallow dissolution.”
This phenomenon, driven by biological factors such as predation, particle aggregation, and microbial respiration, significantly alters ocean chemistry. Despite its importance, shallow dissolution remains largely missing from the major Earth System Models (e.g., CMIP6) that guide global climate predictions.
Diversity among plankton groups and vulnerabilities
The study highlights the unique traits of different calcifying plankton groups, which determine their geographic distribution, ecological role, and vulnerabilities. Coccolithophores, the main producers of CaCO₃, are especially sensitive to acidification, as they lack specialized pumps to remove acidity from their cells.

Foraminifers and pteropods do, but they face different pressures, from oxygen loss to warming waters. Together, these groups shape the fate of carbon in the ocean. Ignoring their diversity risks oversimplifying how the ocean responds to climate stressors.
Toward better climate modeling and prediction
The paper calls for urgent efforts to better quantify group-specific production, dissolution, and export of calcium carbonate, and to incorporate these dynamics into climate models. Doing so would allow for more accurate projections of ocean–atmosphere feedbacks, carbon sequestration, and even the interpretation of sediment records used to reconstruct past climates.
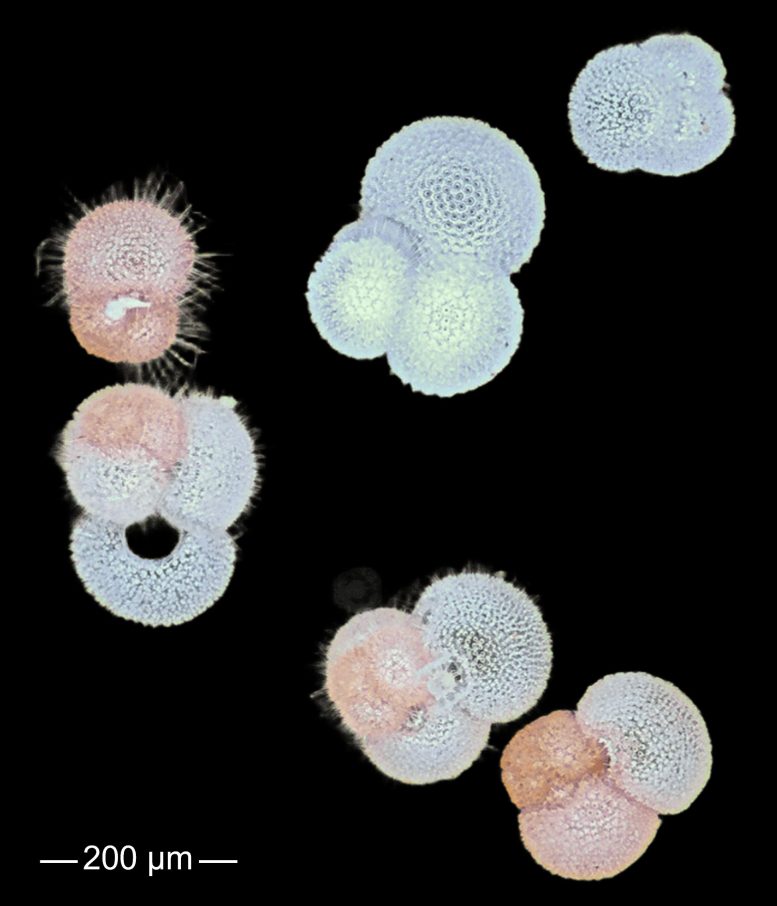
“If we ignore the ocean’s smallest organisms, we might miss important climate dynamics,” says Dr. Ziveri. “Integrating calcifying plankton into climate models could offer sharper predictions and deeper insights into how ecosystems and societies may be affected.”
The researchers conclude that addressing these knowledge gaps is critical to developing a new generation of climate models that better capture the biological complexity of the oceans.

Reference: “Calcifying plankton: From biomineralization to global change” by Patrizia Ziveri, Gerald Langer, Sonia Chaabane, Joost de Vries, William Robert Gray, Nina Keul, Ian A. Hatton, Clara Manno, Richard Norris, Sven Pallacks, Jeremy R. Young, Ralf Schiebel, Stergios Zarkogiannis, Griselda Anglada-Ortiz, Stefania Bianco, Thibault de Garidel-Thoron, Michaël Grelaud, Arturo Lucas, Ian Probert and P. Graham Mortyn, 23 October 2025, Science.
DOI: 10.1126/science.adq8520
Never miss a breakthrough: Join the SciTechDaily newsletter.
Follow us on Google and Google News.
13 Comments
The author implies this is a mitigating force to climate change. It’s not! It’s an aggravating force! I consider the article a lie by omission.
The author implies this is a mitigating force to climate change. It’s not! It’s an aggravating force! He also states that Plankton or not included in climate models. That’s laughable. All climate models include plankton. I consider the article a lie by omission.
The author implies this is a mitigating force to climate change. It’s not! It’s an aggravating force! He also states that Plankton are not included in climate models. That’s laughable. All climate models include plankton. The headline implies there’s some large factor science has missed and it could overturn our whole understanding of climate change and and possibly solve the whole problem. I consider that implication an attempt at a bold face lie.
On November 6th you replied to me saying, “You can’t see any of that detail from the Copernicus satellite I’m calling you out! Debate me on the subject. I don’t think you have the testicles for it.” I’m still waiting for your apology and to have you engage in a debate. It seems that the only thing that you can do is insult people and make unsupported assertions. Saying the same thing three times doesn’t make it any more persuasive.
Of course they’re wrong. That’s why they need to backtrack them all the time.
💯
“This phenomenon, driven by biological factors such as predation, particle aggregation, and microbial respiration, significantly alters ocean chemistry.”
After making this statement, I think that it would have been appropriate to show the Bjerrum Plot for carbonate systems [ https://en.wikipedia.org/wiki/Bjerrum_plot ] that shows how the chemical buffering in the oceans actually works to stabilize the pH. Note that the recent pH has been around 8. We aren’t certain about history because the researchers have largely ignored the sparse samples and commonly rely on a computer model for a claimed pre-industrial pH of 8.2, thus providing a sense of support for the claim of so-called ‘ocean acidification.’
However, I’d like to point out that no less than the renowned Stanford geochemist Konrad Krauskopf has stated in his textbook that, because of the carbonate/borate chemical buffering systems, it is unlikely that the oceans will ever even reach a pH of 7 (neutral) except in stagnant bottom pools enriched in hydrogen sulfide; inorganic solutions of water are not considered “acidic” until the pH is less than 7.
Climate scientists have Not said the oceans would be acidic. But decreasing pH can still harm corals and other shelled creatures. pH under 8 is not healthy for corals.
——————————–
Not plankton but still important
Warming seas threaten key phytoplankton species that fuels the food web, study finds
“For decades, scientists believed Prochlorococcus, the smallest and most abundant phytoplankton on Earth, would thrive in a warmer world. But new research suggests the microscopic bacterium, which forms the foundation of the marine food web and helps regulate the planet’s climate, will decline sharply as seas heat up.
Prochlorococcus inhabit up to 75% of Earth’s sunlit surface waters and produce about one-fifth of the planet’s oxygen through photosynthesis. More crucially, Ribalet said, they convert sunlight and carbon dioxide into food at the base of the marine ecosystem.” ”
AP News
“Climate scientists have Not said the oceans would be acidic.”
Au contraire! The term “ocean acidification,” adopted by alarmist climatologists and many oceanographers, implies that the oceans are on a path that Krauskopf says will never happen. When anyone says that the oceans are becoming MORE acidic, which one sees frequently, it also implies that the oceans are already acidic and becoming more so. Such duplicitous re-branding of the meaning of words results in contradictory claims and confusion. The problem is that many who play in the climatology field have weak geochemistry backgrounds and appear to be unfamiliar with the chemical buffering that stabilizes the pH of oceans.
“pH under 8 is not healthy for corals.” [no citation]
When I searched for “pH tolerance range of tropical corals,” ‘Search Assistant’ came back with, “Tropical corals generally THRIVE in a pH range of about 7.8 to 8.5.” Inasmuch as the carbonate/borate buffering system makes a pH as low as 7.7 unlikely, and the mid-point of that range is about 8.15, it appears that your claim that I quoted is not supported. It is generally accepted that the open ocean pH is currently about 8.1, with some notable diurnal variation in shallow waters. Thus, ocean waters are currently near the mid-point of their optimum range, with the possibility that the open ocean may have been 0.1 pH unit higher a century ago; however, that is problematic as actual measurements have been ignored in favor of the results of a model of unknown veracity. It is rare to see uncertainties associated with ocean pH measurements; however, taking the available numbers at face value, the current pH is only about 0.05 units below the optimum mid-point, which is about 0.05 units lower than what it was presumed to be in pre-industrial times. That is, the stress on tropical corals is no greater today than it was more than a century ago.
You will have to do better if you want have credibility with readers.
“But new research suggests the microscopic bacterium, which forms the foundation of the marine food web and helps regulate the planet’s climate, will decline sharply as seas heat up.”
The unstated assumptions are that if the abundance of the current dominant strain of Prochlorococcus declines, the species will not adapt by reproductively favoring the individuals that are more heat tolerant, or that other species won’t take over the vacated ecological niche. One has to look at the whole picture and not focus on just the possibilities that are supportive of catastrophism.
” Coccolithophores, the main producers of CaCO₃, are especially sensitive to acidification, as they lack specialized pumps to remove acidity from their cells., the main producers of CaCO₃, are especially sensitive to acidification, as they lack specialized pumps to remove acidity from their cells.”
I think that could be stated more clearly and accurately by observing that most calcifiers have the ability to use proton pumps to manipulate the pH at the surface of their shells, thus giving them the ability to repair damage and to optimize the pH at growth surfaces to thereby tolerate a wider range of environmental pH changes.
I was unaware that Coccolithophores lacked the ability to manipulate the pH as other calcifiers do. It is something that I need to look into. I suspect that, like other calcifiers, they rely on something like mucous to insulate their shells from direct contact with the sea water. I’m appreciate hearing from any marine biologists who can enlighten me on what happens with Coccolith’s when they encounter corrosive waters, which also varies with temperature.
Not to mention the absence of discussion regarding the action of volcanoes, or the influence of the ingestion of microscopic creatures by whales, not to mention the whales’ effluent on the condition of the oceanic environment. Where is the real science being done?