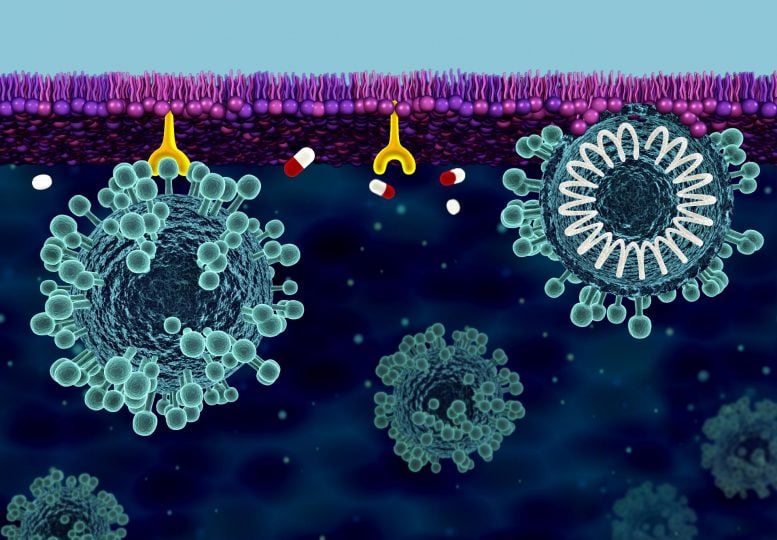
The cell membrane is the cell’s first line of defense against the coronavirus responsible for COVID-19. The surface of the virus is covered with spike proteins, which bind to the cell membrane and facilitate viral infection. Researchers are investigating how this process works and what treatments can help to stop it. Credit: ORNL/Jill Hemman
The cell membrane is the cell’s outermost line of defense against SARS-CoV-2, the novel coronavirus responsible for COVID-19 disease.
Cell membranes are just a few nanometers thick but are essential for life. They serve as a barrier between the cell’s interior and its surrounding environment and host many activities necessary for cell function.
Researchers from Virginia Tech and the Department of Energy’s (DOE’s) Oak Ridge National Laboratory (ORNL) are using neutron scattering to investigate how the cell membrane and the virus impact each other and what therapeutic candidates could make cell membranes more resistant to viral entry. Such information could help experts design strategies for slowing down the progress of viral infection and reducing its harmful effects.
“Developing treatments that interfere with the viral infection process could help reduce the severity of the COVID-19 disease and enable people to recover more quickly,” said John Katsaras, a biophysicist and neutron scattering scientist at ORNL. This, in turn, could decrease the number of hospitalizations and reduce the risk of overwhelming medical facilities.
Membrane-focused mission
The coronavirus hijacks human cells with the help of spike proteins that protrude from its own membrane layer, giving the virus its crown-like appearance. The spike protein attaches to the cell surface and helps merge the viral and cellular membranes. Once the membranes are fused, the virus can enter the cell and create copies of itself, spreading the infection throughout the body.
By determining how the coronavirus penetrates the cell membrane, scientists can develop treatments that hinder this process. Many researchers are exploring ways to combat the virus by targeting its spike proteins, but fewer are paying attention to the site where the infection process begins: the cell membrane.
“For a virus to be active it has to go through the cell membrane,” said Rana Ashkar, an assistant professor in physics at Virginia Tech and former ORNL Shull Fellow who is leading the research effort. “Much of the work that has been done is focused on inactivating the spike protein itself. However, we want to understand how the spike protein interacts with the membrane and what treatments could indirectly block this interaction by targeting the properties of the membrane.”
Ashkar and her research group study the structure and dynamics of biological membrane models to gain insight into cell membrane function. When COVID-19 began to spread, she harnessed her lab’s resources to help the scientific community better understand coronavirus infection.
They collaborated with researchers at ORNL to establish a molecular understanding of the membrane properties that allow viral entry, how membranes change when in contact with the virus, and what membrane modifications could inhibit the infection process.
“ORNL has an international leadership role in analyzing the nanoscopic structure of biological membranes. Because of our expertise, network of collaborators, and neutron scattering capabilities, we were able to quickly pivot our research focus toward better understanding how the coronavirus, and certain other compounds, impact membranes,” said Katsaras.
This project also tapped into the wide range of expertise within the laboratory. “At ORNL we are able to work with experts from many fields, such as physics, chemistry, and biology,” said Jessy Labbé, a cellular and molecular fungal geneticist in the ORNL Biosciences Division. “When the crisis began, we applied this combined knowledge to develop research projects that address some of the biggest challenges related to the pandemic.”
Neutron vision
The team is using the liquids reflectometer (LIQREF) at ORNL’s Spallation Neutron Source (SNS) to examine the conformation of membranes and viral spike proteins, as well as the effects of certain therapeutic candidates. With the instrument, scientists can measure the trajectories of neutrons as they interact with different biological materials. They then use this information to determine how a sample is organized at the molecular level.
“Using this technique, we can capture the membrane’s structure and assess what molecular details change, under physiological conditions, when a membrane is in contact with spike proteins or therapeutic compounds,” said Minh Phan, a postdoctoral research associate at ORNL.
Biological membranes have a nanoscopic profile, making it challenging for researchers to investigate their structures and study how they behave, according to Ashkar. Neutrons, however, are capable of probing biological materials with high resolution without impairing them. Neutron scattering is one of the few techniques available for exploring how the virus and therapeutic candidates interact with membranes on the nanoscale, she said.
Neutrons are also ideal for this study because they can differentiate between hydrogen and its isotopes, such as deuterium. “By selectively replacing hydrogen atoms in membrane samples with deuterium atoms, we have this ability to change the way that neutrons ‘see’ things, so that we can amplify certain aspects and completely obscure others,” said Ashkar.
“As we change the levels of hydrogen and deuterium in our samples, we can pinpoint different features within membranes,” added ORNL instrument scientist John Ankner. “By compiling all these measurements, we can then create one structural model, like putting together pieces of a jigsaw puzzle.”
Building a molecular picture
The researchers performed their experiments with a membrane model that closely mirrors the shape and composition of cell membranes within human lungs, where respiratory viral infections primarily take place. The samples were prepared by Mary Odom, scientific associate at SNS. Using LIQREF, the team first characterized the membrane’s original structure. They then measured how the membrane’s properties change when exposed to either melatonin or azithromycin — commonly available products that are currently being investigated by medical experts as possible treatments for mitigating COVID-19 symptoms.
Melatonin is a natural hormone produced by the brain and plays a role in regulating sleep cycles. Synthetically made melatonin supplements are primarily used for jet lag and sleep disorders but have also been used for alleviating symptoms of various infections. Azithromycin is an antibiotic prescribed to treat a wide range of infections, such as bronchitis and pneumonia.
Ashkar is leading additional characterization experiments at Virginia Tech to further examine how these products affect membrane organization. The initial results suggest that membranes pack more closely together when introduced to melatonin or azithromycin. This change could potentially make membrane intrusions more challenging for viral spike proteins.
The team will incorporate viral spike proteins into the membrane samples and analyze various factors of the protein-membrane complex. These include how the spike protein binds to the membrane, the protein’s insertion mechanism, and the membrane’s response to the protein, such as changes to its compression or stiffness. The team will then examine whether these interactions are disrupted in the presence of the therapeutic candidates.
Neutron scattering efforts have been implemented primarily by Ankner and Phan. Additionally, Jacob Kinnun, an ORNL postdoctoral research associate, conducted complementary experiments to explore how the two compounds affect the molecular packing and binding dynamics of membrane samples.
Pat Collier, a staff researcher at ORNL’s Center for Nanophase Materials Sciences, studied membranes formed by lipid-coated water droplets to gain further information on membrane properties.
Going forward, the researchers envision these methods could be used to establish a platform that rapidly and systemically screens various treatments for their potential to help mitigate both the current pandemic and other viral respiratory threats that may arise in the future.
This work received support through the ORNL Laboratory Directed Research and Development program.






Be the first to comment on "Building a Cell Membrane Defense Against COVID-19"