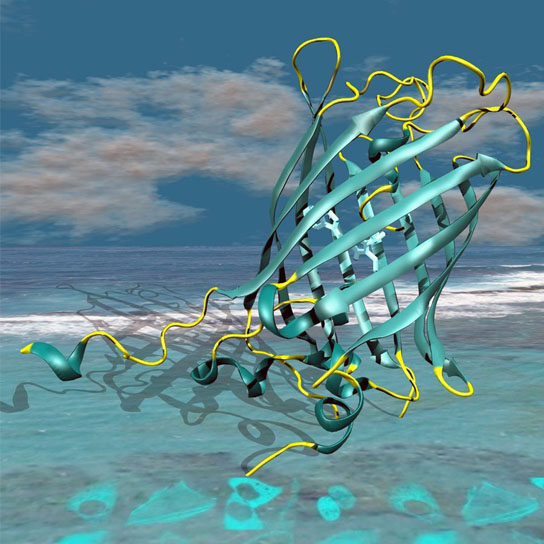
By first uncovering how CFPs store incoming energy and retransmit it as fluorescent light, scientists were able to understand the function of individual atoms within CFPs and pinpoint the part of the molecule that needed to be modified to increase the fluorescence yield, resulting a new CFP called mTurquoise2 that has a fluorescence efficiency of 93%.
Scientists have designed a molecule that emits turquoise light more efficiently than ever seen before in living cells. This improves the sensitivity of cellular imaging, a technique for high-resolution imaging of biological processes inside a living cell. The results have been published in the journal Nature Communications on March 20, 2012.
The lead author of the publication is Antoine Royant from the Institut de Biologie Structurale/CNRS-CEA-Université Joseph Fourier in Grenoble. The team also comprised scientists from the Universities of Amsterdam and Oxford, and the European Synchrotron Radiation Facility (ESRF) in Grenoble.
Cyan fluorescent proteins (CFPs) are very popular in cell biology where they are used to make processes visible inside a living cell, like in a movie. These processes include the changes in the shape of large biological molecules. Since the early 1990s, fluorescent proteins have become one of the most important tools used in the biosciences and have helped the observation of previously invisible processes such as the development of nerve cells in the brain or the spread of cancer cells. The 2008 Nobel Prize in Chemistry crowned their discovery and rapid development.
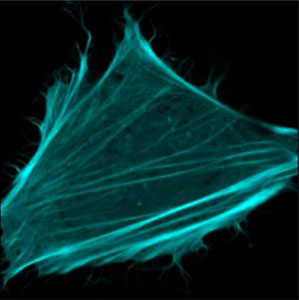
CFPs allow the mapping of many processes in living cells when they can be attached to a protein involved in an interaction or a conformational change. The CFP inside the cell is localized by illuminating the cell with blue light which makes the fluorescent protein emit light of a characteristic color, which is cyan for CFPs. The emission of light from the CFP reveals the target of observation, the protein to which it is attached. However, these molecules have long suffered from a weak fluorescence level, converting merely 36% of the incoming blue into cyan light.
To achieve higher brightness, and with it improved sensitivity of fluorescent imaging, the scientists based in France, led by Antoine Royant, teamed up with colleagues from the Netherlands and the United Kingdom.
First, using highly brilliant X-ray beams at the ESRF, the teams from Grenoble and Oxford uncovered subtle details of how CFPs store incoming energy and retransmit it as fluorescent light: they produced tiny crystals of many different improved CFPs and resolved their molecular structures. These structures revealed a subtle process near the so-called chromophore, the light-emitting complex inside the CFPs, whose fluorescence efficiency could be modulated by the environment. “We could understand the function of individual atoms within CFPs and pinpoint the part of the molecule that needed to be modified to increase the fluorescence yield,” says David von Stetten from the ESRF.
In parallel to this work, the Amsterdam team led by Theodorus Gadella used an innovative screening technique to study hundreds of modified CFP molecules, measuring their fluorescence lifetimes under the microscope to identify which had improved properties.
The result of this rational design is a new CFP, called mTurquoise2. By combining structural and cellular biology efforts, the researchers managed to show that mTurquoise2 has a fluorescence efficiency of 93%, unmatched for this type of protein.
The new molecule will allow life scientists to study protein-protein interactions in living cells with unprecedented sensitivity. High sensitivity matters in processes where only a few proteins are involved and signals are weak, and in fast reactions where the time available for accumulating fluorescent light is short.
“With the new protein, many studies can now be performed with levels of accuracy and detail that were impossible yesterday. Moreover, thanks to this novel approach taking into account the structural dynamics of the protein, scientists now hope to design improved fluorescent proteins emitting light of different colors for use in other applications,” concludes Antoine Royant.
Reference: “Structure-guided evolution of cyan fluorescent proteins towards a quantum yield of 93%” by Joachim Goedhart, David von Stetten, Marjolaine Noirclerc-Savoye, Mickaël Lelimousin, Linda Joosen, Mark A. Hink, Laura van Weeren, Theodorus W.J. Gadella Jr and Antoine Royant, 20 March 2012, Nature Communications.
DOI: 10.1038/ncomms1738
Never miss a breakthrough: Join the SciTechDaily newsletter.
Follow us on Google and Google News.