
Ocean acidification is making it harder for sea butterflies to build shells, threatening their survival and disrupting Antarctic marine ecosystems.
The oceans are becoming more acidic because of the rapid release of carbon dioxide (CO2) caused by anthropogenic (human) activities, such as burning of fossil fuels. So far, the oceans have taken up around 30% of all anthropogenic CO2 released into the atmosphere. The continuous increase of CO2 has a substantial effect on ocean chemistry because CO2 reacts with water and carbonate molecules. This process, called ‘ocean acidification’, lowers pH, and calcium carbonate becomes less available. This is a problem for calcifying organisms, such as corals and mollusks, that use calcium carbonate as the main building blocks of their exoskeleton.
In particular, organisms that build their shells from a type of calcium carbonate known as ‘aragonite’ are in trouble because aragonite is extremely soluble in seawater. Sea butterflies, tiny, swimming sea snails, build their shells of aragonite. Therefore, they are also known as ‘the canaries of the coalmine’ because they are expected to be amongst the first organisms to be affected by ocean acidification.
The Southern Ocean, around the Antarctic continent, is a region of high concern regarding ocean acidification. Globally, this region will experience acidified conditions first, because colder waters take up CO2 more readily. This is comparable to soda: you will find more bubbles dissolved in your soda when it is cold. Already within several decades, aragonite will be scarce in the Southern Ocean, posing a large problem for local sea life, such as sea butterflies.
Large numbers of sea butterflies inhabit the Southern Ocean and function as an important component of the food web where they are eaten by fish, seabirds and even whales. “It is important to gain a deeper understanding of the impacts of an increasingly acidifying ocean on the shell growth of sea butterflies”, says Lisette Mekkes, PhD student at Naturalis Biodiversity Center and the University of Amsterdam. “Imagine that a highly abundant sea butterfly, Limacina retroversa, disappears from this region; that would have major implications for the rest of local sea life that depend on them for food, and for the calcium carbonate export from surface waters to the deep sea.”

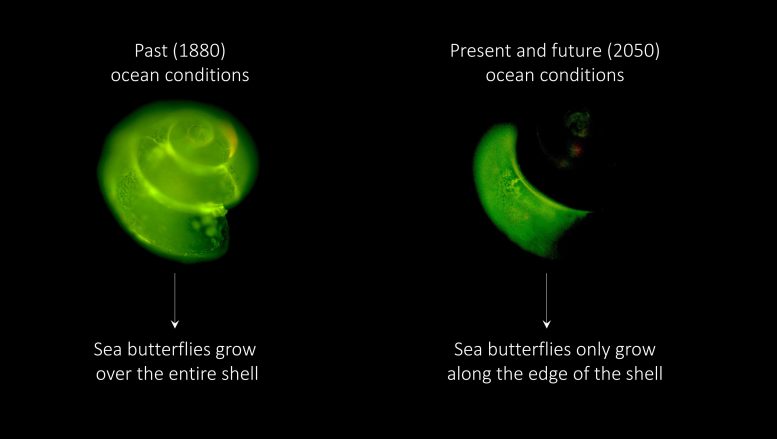
As part of Lisette’s PhD research, sea butterflies from the Southern Ocean were exposed to different ocean conditions comparable to the past, the present and the near-future. Through a green, fluorescing substance, shell growth of the sea butterflies was visualized. Based on this fluorescing, the scientists discovered that sea butterflies already experience difficulties in building their shells in today’s Southern Ocean. This will become even more difficult in the upcoming decades.
The Decline in Shell Quality and Density
In past conditions, equivalent to the year 1880 (before an increased concentration of CO2 was absorbed by the oceans) sea butterflies were able to calcify over their entire shell: they built thicker and larger shells. In the present-day and future ocean conditions, sea butterflies mostly built more shell material only along the edge of the shell. Moreover, shells exposed to the future conditions did not increase in shell weight and had a lower density compared to past and present conditions, suggesting that calcification will be further compromised in the future.
“It appears that sea butterflies change their calcification strategy as a consequence of ocean acidification” explains Katja Peijnenburg, group leader at Naturalis Biodiversity Center and the University of Amsterdam. “As long as the oceans are not acidifying, sea butterflies can invest in growing thicker and larger shells. However, when the oceans acidify and aragonite is less available, they invest mostly in becoming larger” concludes Peijnenburg.
Facing an Uncertain Future in Acidifying Oceans
Although it is generally known that sea butterflies are vulnerable to changes in ocean chemistry, it is alarming to find out that they are already experiencing difficulties building their shells in today’s ocean. The scientists wonder how much longer sea butterflies will be able to build their shells as the global ocean continues to acidify.
Reference: “Effects of Ocean Acidification on Calcification of the Sub-Antarctic Pteropod Limacina retroversa” by Lisette Mekkes, Guadalupe Sepúlveda-Rodríguez, Gintarė Bielkinaitė, Deborah Wall-Palmer, Geert-Jan A. Brummer, Linda K. Dämmer, Jef Huisman, Emiel van Loon, Willem Renema and Katja T. C. A. Peijnenburg, 2 March 2021, Frontiers in Marine Science.
DOI: 10.3389/fmars.2021.581432
Never miss a breakthrough: Join the SciTechDaily newsletter.
Follow us on Google and Google News.
3 Comments
“The oceans are becoming more acidic because of the rapid release of carbon dioxide (CO2) caused by anthropogenic (human) activities, …”
It is only grammatically correct to say that someone is becoming more angry if they are already angry. Similarly, something can only become “more acidic” if it is already acidic. Chemists define a pH of 7 as the boundary between acidic and alkaline aqueous solutions. The article notes that all the pH values being discussed are greater than 7, and thus alkaline!
“… aragonite is extremely soluble in seawater.”
Aragonite is more soluble than calcite, the other common carbonate found in shells. However, calling aragonite “extremely soluble” is hyperbole and inaccurate. Table salt is “extremely soluble.”
From the actual research paper, the authors remark about the “rapidly acidifying waters of the sub-Antarctic.” Typically, the diurnal and seasonal changes in ocean pH are much greater than the assumed average change during the latter part of the industrial revolution (claimed to have decreased from 8.19 to 8.06, currently, where the uncertainty is stated to be at least 0.01).
All calcifiers have to contend with changing pH, particularly those that live in upwelling zones. They have various strategies to cope, including the use of mucous and chitin to protect their shells from conditions that might dissolve them. The research article talks about removing organic material from the shells. They may have destroyed some important information about how, and to what degree, the pteropods were coping with the experiment environment.
See my critique of the poor practice of referring to decreasing pH as “ocean acidification”:
http://wattsupwiththat.com/2015/09/15/are-the-oceans-becoming-more-acidic/
An excellent critique, Clyde Spencer!
We are killing everything.