
Scientists reveal how an intranasally delivered oxytocin derivative could be used to treat Alzheimer’s disease.
Alzheimer’s disease (AD), characterized by an accumulation of ß-amyloid protein (Aß) in brain tissue, is a leading cause of dementia. Researchers at Tokyo University of Science have previously reported on the oxytocin-induced reversal of impaired synaptic plasticity triggered by amyloid ß peptide (25-35) (Aß25-35). They now show that an oxytocin derivative with modifications to enhance brain perfusion can reverse Aß25-35-induced cognitive impairment in mice.
The cognitive decline and memory loss observed in Alzheimer’s disease (AD) is attributed to the accumulation of ß-amyloid protein (Aß), which impairs neural function in the brain. Experimentation has shown that oxytocin, a peptide hormone primarily responsible for parturition, bonding, and lactation, also regulates cognitive behavior in the rodent central nervous system (CNS). This finding, along with the identification of oxytocin receptors in CNS neurons, has spurred interest in the potential role of oxytocin in reversing memory loss tied to cognitive disorders like AD.
Overcoming Blood-Brain Barrier Challenges with PAS-CPPs-Oxytocin
However, peptides like oxytocin are characterized by weak blood-brain barrier permeability, and so can only be efficiently delivered to the brain via intracerebroventricular (ICV) administration. ICV, however, is an invasive technique that is impractical to implement clinically.
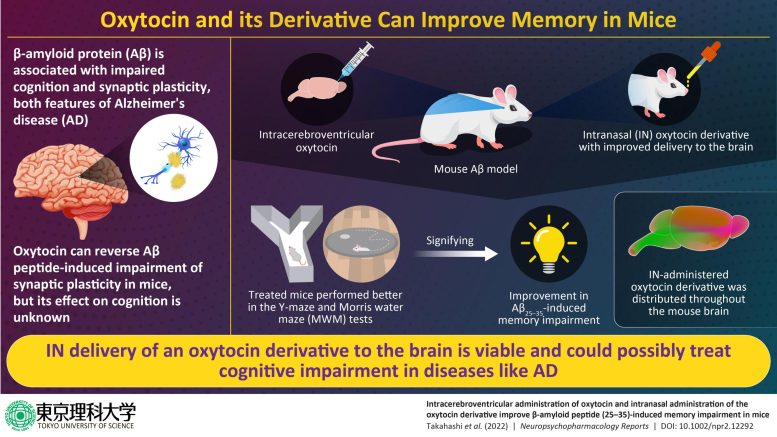
Delivering peptides to the CNS via intranasal (IN) administration is a viable clinical option. Prof. Chikamasa Yamashita at Tokyo University of Science recently patented a method to increase the efficiency of peptide delivery to the brain, by introducing cell-penetrating peptides (CPPs) and a penetration-accelerating sequence (PAS) through structural modifications. Previous work had confirmed that both CPPs and the PAS benefit the nose-to-brain delivery pathway. Now, a group of researchers, led by Prof. Akiyoshi Saitoh and Prof. Jun-Ichiro Oka, leveraged this approach to prepare an oxytocin derivative: PAS-CPPs-oxytocin. Their findings were recently published online in Neuropsychopharmacology Reports.
“We have previously shown that oxytocin reverses amyloid 𝛽 peptide (25-35) (A𝛽25-35)-induced impairment of synaptic plasticity in rodents. We wanted to see if PAS-CPPs-oxytocin could be delivered more efficiently to the mouse brain for clinical application, and if it improved cognitive functional behavior in mice,” states Prof. Oka.
The group first developed an A𝛽25-35 peptide-induced amnesia model by supplying A𝛽25-35 to the mouse brain using ICV delivery. During the course of the study, the spatial working and spatial reference memories of these mice were evaluated using the Y-maze and Morris water maze (MWM) tests. After confirming that memory was affected in A𝛽25-35-impaired mice, PAS-CPPs-oxytocin and native oxytocin were administered using the IN and ICV routes respectively, to see if learning and memory improved in the treated mice. Finally, the distribution of the IN-administered oxytocin derivative in brain tissue was profiled by imaging of a fluorescent-tagged oxytocin derivative.
Clinical Potential of Intranasal Oxytocin Derivative for Alzheimer’s Treatment
The results of this study were quite promising! The tagged PAS-CPPs-oxytocin showed distribution throughout the mouse brain following its IN administration. While the ICV administration of native oxytocin improved test outcomes in both the Y-maze and MWM tests, the IN-administered PAS-CPPs-oxytocin yielded memory-improving effects in the Y-maze test. Hailing the team’s discovery, Prof. Oka says, “My team is the first to show that the oxytocin derivative can improve the A𝛽25-35-induced memory impairment in mice. This suggests that oxytocin may help reduce the cognitive decline we see in Alzheimer’s disease.”
Why are these findings clinically useful? Prof. Oka explains the broader implications of their work, “The oxytocin derivative enters the brain more efficiently. Furthermore, since IN delivery is a non-invasive procedure, this modified version of the hormone could potentially be a clinically viable treatment for Alzheimer’s disease.”
Reference: “Intracerebroventricular administration of oxytocin and intranasal administration of the oxytocin derivative improve ß-amyloid peptide (25–35)-induced memory impairment in mice” by Junpei Takahashi, Yudai Ueta, Daisuke Yamada, Sachie Sasaki-Hamada, Takashi Iwai, Tomomi Akita, Chikamasa Yamashita, Akiyoshi Saitoh and Jun-Ichiro Oka, 19 September 2022, Neuropsychopharmacology Reports.
DOI: 10.1002/npr2.12292
Never miss a breakthrough: Join the SciTechDaily newsletter.
Follow us on Google and Google News.
3 Comments
THAT COULD BE A TRUE BREAKTHRU IN THAT FIELD, SOMETHING SEEMS TO HAVE BEEN VERY ELUSIVE BUT MUCH NEEDED TO HELP PEOPLE AFFLICTED WITH THAT TERRIBLE DISEASE..I HAVE LOST SEVERAL. RELATIVES TO THEM BOTH..BRAVO TO THAT COURAGEOUS TEAM
Maybe I misunderstood. It sounds like you’re saying Alzheimer’s is a sort of cataract on the brain. Berries are good for plaque buildup in the veins, thrombosis, seems they should help for Alzheimer’s as well
How to get this treatment,A blessing 🙏