
An analysis of 20,000 people’s data from the UK Biobank reveals that diabetes accelerates the normal process of brain aging, with longer diabetes duration associated with higher neurodegeneration
According to a study recently published in eLife, researchers have shown that type 2 diabetes patients’ natural brain aging is accelerated by around 26% compared to people without the condition.
The scientists looked at the association between normal brain aging and type 2 diabetes and discovered that type 2 diabetes has a similar pattern of neurodegeneration as aging but proceeds quicker. This study has many significant implications, one of which is that even normal brain aging may be linked with changes in the brain’s regulation of glucose by insulin.
The findings also imply that there could already be severe structural damage to the brain by the time type 2 diabetes is formally identified. Therefore, it is imperative to develop sensitive methods to identify brain changes caused by diabetes.
Although there is considerable evidence associating type 2 diabetes with cognitive deterioration, few patients today get a full cognitive assessment as part of their clinical care. It might be difficult to discern between normal brain aging that occurs in middle life and brain aging caused or hastened by diabetes. To date, no studies have directly compared neurological changes in healthy people throughout the course of their lives to those seen by people of the same age with diabetes.
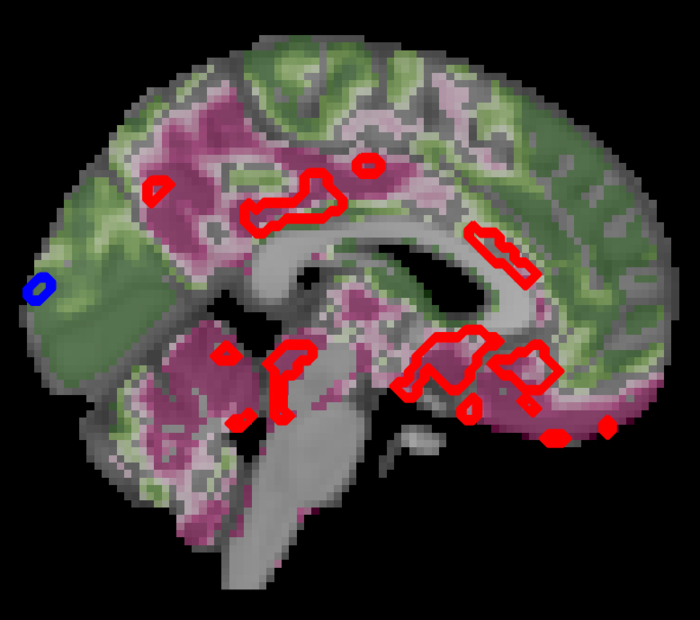
“Routine clinical assessments for diagnosing diabetes typically focus on blood glucose, insulin levels, and body mass percentage,” says first author Botond Antal, a Ph.D. student at the Department of Biomedical Engineering, Stony Brook University, New York, US. “However, the neurological effects of type 2 diabetes may reveal themselves many years before they can be detected by standard measures, so by the time type 2 diabetes is diagnosed by conventional tests, patients may have already sustained irreversible brain damage.”
To define the impact of diabetes on the brain over and above normal aging, the team made use of the largest available brain structure and function dataset across human lifespan: UK Biobank data from 20,000 people aged 50 to 80 years old. This dataset includes brain scans and brain function measurements and holds data for both healthy individuals and those with a type 2 diabetes diagnosis. They used this to determine which brain and cognitive changes are specific to diabetes, rather than just aging, and then confirmed these results by comparing them with a meta-analysis of nearly 100 other studies.
Cognitive and Structural Brain Changes Linked to Diabetes
Their analysis showed that both aging and type 2 diabetes cause changes in executive functions such as working memory, learning, and flexible thinking, and changes in brain processing speed. However, people with diabetes had a further 13.1% decrease in executive function beyond age-related effects, and their processing speed decreased by a further 6.7% compared to people of the same age without diabetes. Their meta-analysis of other studies also confirmed this finding: people with type 2 diabetes had consistently and markedly lower cognitive performance compared to healthy individuals who were the same age and similarly educated.
The team also compared brain structure and activity between people with and without diabetes using MRI scans. Here, they found a decrease in grey brain matter with age, mostly in a region called the ventral striatum – which is critical to the brain’s executive functions. Yet people with diabetes had even more pronounced decreases in gray matter beyond the typical age-related effects – a further 6.2% decrease in grey matter in the ventral striatum, but also loss of grey matter in other regions, compared with normal aging.
Accelerated Brain Aging and Need for Early Detection
Together, the results suggest that the patterns of type 2 diabetes-related neurodegeneration strongly overlap with those of normal aging, but that neurodegeneration is accelerated. Moreover, these effects on brain function were more severe with increased duration of diabetes. In fact, the progression of diabetes was linked with a 26% acceleration of brain aging.
“Our findings suggest that type 2 diabetes and its progression may be associated with accelerated brain aging, potentially due to compromised energy availability causing significant changes to brain structure and function,” concludes senior author Lilianne Mujica-Parodi, Director of the Laboratory for Computational Neurodiagnostics, Stony Brook University. “By the time diabetes is formally diagnosed, this damage may already have occurred. But brain imaging could provide a clinically valuable metric for identifying and monitoring these neurocognitive effects associated with diabetes. Our results underscore the need for research into brain-based biomarkers for type 2 diabetes and treatment strategies that specifically target its neurocognitive effects.”
The study was funded by the W. M. Keck Foundation, the National Science Foundation, and the Baszucki Brain Research Fund.
Reference: “Type 2 diabetes mellitus accelerates brain aging and cognitive decline: Complementary findings from UK Biobank and meta-analyses” by Botond Antal, Liam P McMahon, Syed Fahad Sultan, Andrew Lithen, Deborah J Wexler, Bradford Dickerson, Eva-Maria Ratai and Lilianne R Mujica-Parodi, 24 May 2022, eLife.
DOI: 10.7554/eLife.73138
Never miss a breakthrough: Join the SciTechDaily newsletter.
Follow us on Google and Google News.
1 Comment
Does this consider controlled vs uncontrolled; ie, does control eliminate or reduce the ongoing damage?