Discovery that bacterial biofilms behave like animal embryos sheds new light on the origin of life.
Bacteria are a dominant form of life that inhabit every environment on Earth. This includes human bodies, where they outnumber our cells and genes and regulate our existence for good or bad. Bacteria are regularly viewed as simple, single-celled organisms. As bacteria are ancient, it is widely accepted that a bacteria-like, unicellular being was the first life. Recent work published in ‘Molecular Biology and Evolution’ by an international research team challenges these views.
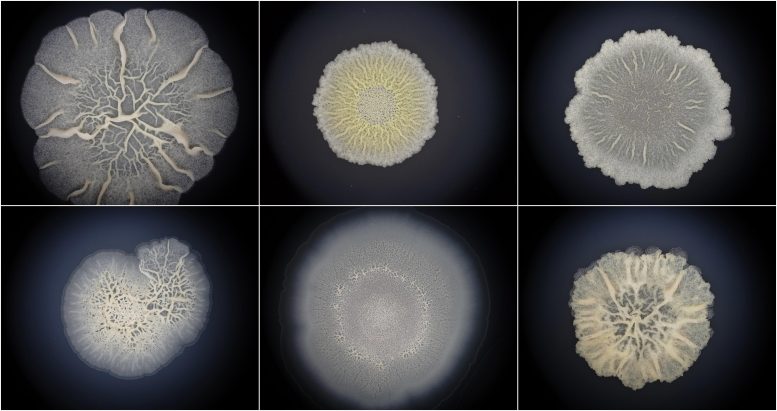
In this paper, the researchers took evolutionary tools to study the growth of biofilms, the most common bacterial lifestyle characterized by the tight clustering of bacterial cells on surfaces. “Surprisingly, we found that the development of bacterial biofilms is comparable to animal embryogenesis. This means that bacteria are true multicellular organisms just like we are. Considering that the oldest known fossils are bacterial biofilms, it is quite likely that the first life was also multicellular, and not a single-celled creature as considered so far,” explains Prof Tomislav Domazet-Lošo from the Rudjer Boskovic Institute and the Catholic University of Croatia in Zagreb who led the research.
Bacteria are the dominant form of life that inhabit every available environment on Earth. This includes human bodies, where they outnumber our cells and genes, and regulate our existence for good and bad. Bacteria are regularly viewed as simple single-celled organisms. As bacteria are ancient, it is also widely accepted that a bacteria-like unicellular being was the first life. However, a recent work published in Molecular Biology and Evolution by an international research team challenges these long-held views. In this study, the researchers took evolutionary tools to study the growth of biofilms, the most common bacterial lifestyle characterized by the tight clustering of bacterial cells on surfaces. Surprisingly, they found that the development of bacterial biofilms is comparable to animal embryogenesis. This means that bacteria are true multicellular organisms just like we are. Considering that the oldest known fossils are bacterial biofilms, it is quite likely that the first life was also multicellular, and not a single-celled creature as considered so far, explain the researchers in their study. The video shows Bacillus growth mirroring the embryonic development. Credit: Dr. Momir Futo, postdoctoral researcher, Laboratory of Evolutionary Genetics, Rudjer Boskovic Institute
Secret life of bacterial biofilms
Macroscopic organisms like animals and plants are never germ-free. They are always accompanied by bacteria and other microorganisms that persist on their inner and outer surfaces. Yet, these symbiotic bacteria are just a tiny fraction of the bacterial diversity that occupy every bit of the biosphere including vast subterranean habitats in the Earth’s crust. In all of these sites bacterial cells are locally organized in morphologically structured clusters known as biofilms. Beside natural habitats, biofilms are also extremely important in industrial and biomedical settings where they often lead to biofouling or persistent infections that are resistant to therapies.
For some time, microbiologist have recognized that bacterial cells live a rich social life in biofilms, however, it has remained obscure if these diverse interactions make a multicellular organism. “Evolutionary methods to study collective behavior of cells in animal development were at hand, but no one tried to transfer this technology from animal embryos to bacterial biofilms. Perhaps people were uncomfortable to challenge the special status of animal multicellularity, the idea that is culturally hardwired,” emphasizes Domazet-Lošo.

Technology translation
Previous work of Domazet-Lošo and his team was focused on evolutionary genomics and animal development. They were able to show that evolution is mirrored in embryos, thus confirming more than 150 years old conjecture that ontogeny parallels phylogeny in animals. To reach these findings, they developed a computational approach, named genomic phylostratigraphy, that allows evolutionary dating of genes and proteins on a large scale. Now together with researchers from University of Zagreb, Chalmers University and Technical University of Denmark they further refined this tool to meet the specifics of bacteria. “We generated the first phylostratigraphic maps of bacteria and this allowed us to link bacterial phenotypes in biofilms to evolutionary information,” continues Domazet-Lošo.
Bacillus biofilms behave like animal embryos
In order to feed their new bioinformatic pipeline, researchers sampled biofilms of Bacillus subtilis, a model organism for biofilm research that inhabits soil as well as human gut. Using transcriptome sequencing and protein quantification they established transcript and protein expression levels for all Bacillus genes during the process of biofilm growth.
“Surprisingly, we found that evolutionary younger genes were increasingly expressed towards the later timepoints of biofilm growth. In other words, we found that Bacillus ontogeny strongly recapitulates phylogeny. So far, these patterns have been considered the signature of embryo development in complex eukaryotes.” stresses Domazet-Lošo. The research team then followed the trail and looked for other features of embryogenesis in biofilms like stage-organized architecture, increased use of multicellularity genes, and molecular links to morphology changes, and to their excitement, they found these properties as well.
Towards the control of biofilms
These findings challenge the special status of eukaryotic multicellularity and show that bacteria are more complex than we thought. The tools developed in this study could also help in finding efficient procedures for biofilm control – a largely unsolved problem in industry and medicine.
“Our results point that a biofilm should be viewed as a multicellular individual, and not as a pile of individual cells. Like in animal embryogenesis, every developmental phase has its own peculiarities. Critical transition stages in biofilm growth could now be targeted via their stage-specific genes that we detected. This could be a game changer in treating biofilm-related diseases, and in preventing industrial losses.” concludes Domazet-Lošo.
Reference: “Embryo-Like Features in Developing Bacillus subtilis Biofilms” by Momir Futo, Luka Opašić, Sara Koska, Nina Čorak, Tin Široki, Vaishnavi Ravikumar, Annika Thorsell, Maša Lenuzzi, Domagoj Kifer, Mirjana Domazet-Lošo, Kristian Vlahoviček, Ivan Mijakovic and Tomislav Domazet-Lošo, 1 September 2020, Molecular Biology and Evolution.
DOI: 10.1093/molbev/msaa217
Never miss a breakthrough: Join the SciTechDaily newsletter.
Follow us on Google and Google News.
2 Comments
Very Interesting.
This is a theory I have been thinking about since yesterday Night (IST). Slept on it for some time and came up with the following Ideas from the Unkown Unconscious Quadrant —only accessible in dream state.
Theory I am putting out into the ether for Medical Researchers and Doctors and Scientists to consider and prove or disprove is as follows:
Complex Eukaryotes = Humans
Human Gut = Second Brain
Friendly,Virus amd Phagocytes AND frieendly Bacteria = Healthy Mortals = L – Amino Acid based ( Chirality) Nucleotides
Unfriendly , Virus and Phagocytes AND Bacteria = Unealthy or sick mortals = D – Amino Acid based ( Chirality) Nucleotides
Not proposing that there needs to be too many D- Amino Acid based Nucleotides. Even a few buried in the polymeric structure which helps the Virus to fit on the receptor of the human organs like a glove can result in sickness and destruction of the organs.
May just be like if Matter and Anti Matter meet it can result in complete anhlation, so also it is possible that when Life and Anti – Life meet , it destroys the critical organs and results in anhilation of Life and leaves behind a lifeless body?
THis is all only a Theory. Upto all the brilliant minds and scientists across the Globe to investigate/
Also , have always wondered why different mammalian species have different ife spans. A Tortoise can live for close to 300 years. A Dog for a dozen. Some humans are long lived and others have much shorter life spans. Matbe the immunity developed over three million years by humans has a part to play in fending of unfriendly visitors , who can enter Human Ecosystems invited or univited through nine openings.
Remember reading, that long lived people consume a lot of curd, which has friendly digestive bacteria and is considered a very important food in India and increasingly many parts of the world. Similarly, many old wives tales learnt through experiance over eons and practised by people who live very long lives may be related to healthy food habits and lifestyles, which may minimize the introduction of unfrienfdly Viruses, bacteria , and other pathogens which increases the risk of illness. The critical systems in Humans for physical, psychological, mental and overall health are the following from a medical perspective. Respiratory, Circulatory, Excretory, Consciousness, Creative and Life Force itself. As long as we keep all of these healthy and in good working condition, we can live along fulfilling life and avoid unnecessary health issues. However, all it takes for all of Humanity to be affected is one intransigent and obstinate idiot who invites such unfriendly , deadly and highly inffective bugger and the rest may be history!
Similarly, the Japanese consume a lot of Seaaweed in their Diet and this may be beneficial for long life spans.
Once again this is a theory and needs to be proved by SCientific studies. Till such time as we know better smart risk managementmay require us as a species changing food habits globally to minimise such likelihood of introducing unfriendly Viruses , Pathogend and Bacteria into the Human Ecosystem which may have lived peacefully in other mamalian and non mamalian ecosystems and when they enter the human ecosystem they may cause havoc not only to the consumer but all o.f humanity as we appear to be watching live.