
A second steroid that has been manufactured by New England Compounding Center has been linked to another potential meningitis infection. The ongoing fungal meningitis outbreak has infected 231 people and killed 16.
NECC was responsible for the 52,848 vials of methylprednisolone acetate, a steroid that is injected to alleviate back pain, which caused the outbreak. A second steroid, triamcinolone acetonide, which is used to treat irritated skin, has also been linked to the infections. There are even concerns that a drug used to paralyze the heart is causing these infections.
All of these drugs from NECC have been recalled since October 6th, but the existence of more tainted drugs could potentially expand the number of people at risk. The exact symptoms take between one and four weeks to present themselves, which could mean that more cases are on the way. This fungal variety of meningitis isn’t contagious, so there’s no risk of the disease spreading.
NECC has also ceased operations. There have been infections in 15 different states. About 14,000 patients that have received methylprednisolone acetate may be at risk for meningitis.
Agents from the FDA have searched the NECC offices. The raid took place as calls came for an even wider probe into whether the company had broken federal laws dealing with controlled substances.
The 16th death was announced this Tuesday, in Virginia. There has been an increase of 19 infections from the previously revealed figures. NECC engaged in what’s called drug compounding, which isn’t regulated by the FDA. The process involves preparing specific doses of approved medications, based on the guidance of a physician to meet a patient’s need.
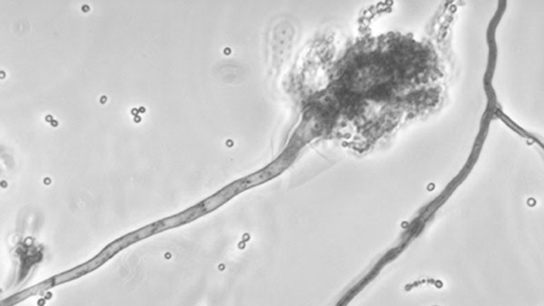
The outbreak has been tied to a type of mold of the genera Aspergillus and Exserohilum. Aspergillus has rarely been associated with fungal meningitis.
Never miss a breakthrough: Join the SciTechDaily newsletter.
Follow us on Google and Google News.