Rice University engineers have created a light-powered nanoparticle that could shrink the carbon footprint of a major segment of the chemical industry.
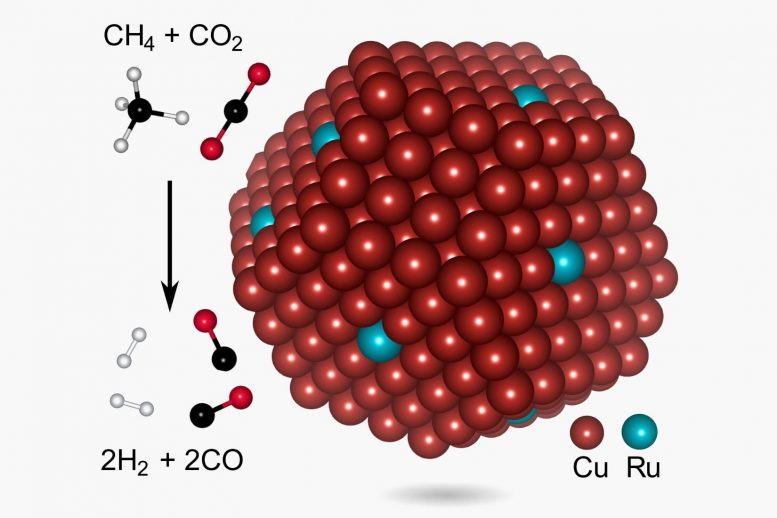
The particle, tiny spheres of copper dotted with single atoms of ruthenium, is the key component in a green process for making syngas, or synthesis gas, a valuable chemical feedstock that’s used to make fuels, fertilizer, and many other products. Researchers from Rice, UCLA, and the University of California, Santa Barbara (UCSB), describe the low-energy, low-temperature syngas production process this week in Nature Energy.
“Syngas can be made in many ways, but one of those, methane dry reforming, is increasingly important because the chemical inputs are methane and carbon dioxide, two potent and problematic greenhouse gases,” said Rice chemist and engineer Naomi Halas, a co-corresponding author on the paper.

Syngas is a mix of carbon monoxide and hydrogen gas that can be made from coal, biomass, natural gas, and other sources. It’s produced at hundreds of gasification plants worldwide and is used to make fuels and chemicals worth more than $46 billion per year, according to a 2017 analysis by BCC Research.
Catalysts, materials that spur reactions between other chemicals, are critical for gasification. Gasification plants typically use steam and catalysts to break apart hydrocarbons. The hydrogen atoms pair up to form hydrogen gas, and the carbon atoms combine with oxygen in the form of carbon monoxide. In dry reforming, the oxygen atoms come from carbon dioxide rather than steam. But dry reforming hasn’t been attractive to industry because it typically requires even higher temperatures and more energy than steam-based methods, said study first author Linan Zhou, a postdoctoral researcher at Rice’s Laboratory for Nanophotonics (LANP).
Halas, who directs LANP, has worked for years to create light-activated nanoparticles that insert energy into chemical reactions with surgical precision. In 2011, her team showed it could boost the amount of short-lived, high-energy electrons called “hot carriers” that are created when light strikes metal, and in 2016 they unveiled the first of several “antenna reactors” that use hot carriers to drive catalysis.
One of these, a copper and ruthenium antenna reactor for making hydrogen from ammonia, was the subject of a 2018 Science paper by Halas, Zhou, and colleagues. Zhou said the syngas catalyst uses a similar design. In each, a copper sphere about 5-10 nanometers in diameter is dotted with ruthenium islands. For the ammonia catalysts, each island contained a few dozen atoms of ruthenium, but Zhou had to shrink these to a single atom for the dry reforming catalyst.

“High efficiency is important for this reaction, but stability is even more important,” Zhou said. “If you tell a person in industry that you have a really efficient catalyst they are going to ask, ‘How long can it last?’”
Zhou said the question is important for producers, because most gasification catalysts are prone to “coking,” a buildup of surface carbon that eventually renders them useless.
“They cannot change the catalyst every day,” Zhou said. “They want something that can last.”
By isolating the active ruthenium sites where carbon is dissociated from hydrogen, Zhou reduced the chances of carbon atoms reacting with one another to form coke and increased the likelihood of them reacting with oxygen to form carbon monoxide.
“But single-atom islands are not enough,” he said. “For stability, you need both single atoms and hot electrons.”
Zhou said the team’s experimental and theoretical investigations point to hot carriers driving hydrogen away from the reactor surface.
“When hydrogen leaves the surface quickly, it’s more likely to form molecular hydrogen,” he said. “It also decreases the possibility of a reaction between hydrogen and oxygen, and leaves the oxygen to react with carbon. That’s how you can control with the hot electron to make sure it doesn’t form coke.”
Halas said the research could pave the way “for sustainable, light-driven, low-temperature, methane-reforming reactions for production of hydrogen on demand.”
“Beyond syngas, the single-atom, antenna-reactor design could be useful in designing energy-efficient catalysts for other applications,” she said.
Reference: “Light-driven methane dry reforming with single atomic site antenna-reactor plasmonic photocatalysts” by Linan Zhou, John Mark P. Martirez, Jordan Finzel, Chao Zhang, Dayne F. Swearer, Shu Tian, Hossein Robatjazi, Minhan Lou, Liangliang Dong, Luke Henderson, Phillip Christopher, Emily A. Carter, Peter Nordlander and Naomi J. Halas, 6 January 2020, Nature Energy.
DOI: 10.1038/s41560-019-0517-9
The technology has been licensed by Syzygy Plasmonics, a Houston-based startup whose co-founders include Halas and study co-author Peter Nordlander.
Halas is Rice’s Stanley C. Moore Professor of Electrical and Computer Engineering and professor of chemistry, bioengineering, physics and astronomy, and materials science and nanoengineering. Nordlander is the Wiess Chair and Professor of Physics and Astronomy, and professor of electrical and computer engineering, and materials science and nanoengineering.
Additional co-authors include Chao Zhang, Dayne Swearer, Shu Tian, Hossein Robatjazi, Minhan Lou, Liangliang Dong and Luke Henderson, all of Rice; John Mark Martirez and Emily Carter, both of UCLA; and Jordan Finzel and Phillip Christopher of UCSB.
The research was supported by the Welch Foundation, the Air Force Office of Scientific Research and the Department of Defense.
Never miss a breakthrough: Join the SciTechDaily newsletter.
Follow us on Google and Google News.