
Scientists have found a special group of microglia (brain immune cells) that can help protect against Alzheimer’s disease.
These protective cells reduce inflammation, slow the buildup of harmful plaques, and may preserve memory and brain function. This discovery not only explains why certain genetic traits reduce Alzheimer’s risk but also opens up the possibility of new treatments that use the brain’s own immune system to fight the disease.
Microglia: The Brain’s Double Agents
In Alzheimer’s disease, which is the most common cause of dementia, microglia (the immune cells of the brain) can behave in very different ways. At times, they help protect the brain, and at other moments, they contribute to damage. Their shifting roles influence how quickly and severely the disease develops.
Researchers from the Icahn School of Medicine at Mount Sinai, together with teams from the Max Planck Institute for Biology and Ageing in Cologne, Germany, The Rockefeller University, The City University of New York, and several global partners, have now identified a specific group of microglia that appears to shield the brain. This finding may guide the development of new treatment strategies for Alzheimer’s disease.
In a study released November 5 in Nature, the scientists report that microglia with lower levels of a transcription factor known as PU.1, and with co-expression of a lymphoid-like receptor called CD28, help reduce inflammation in the brain. These cells also slow the formation of amyloid plaques and the spread of toxic tau proteins, which are central features of Alzheimer’s pathology.
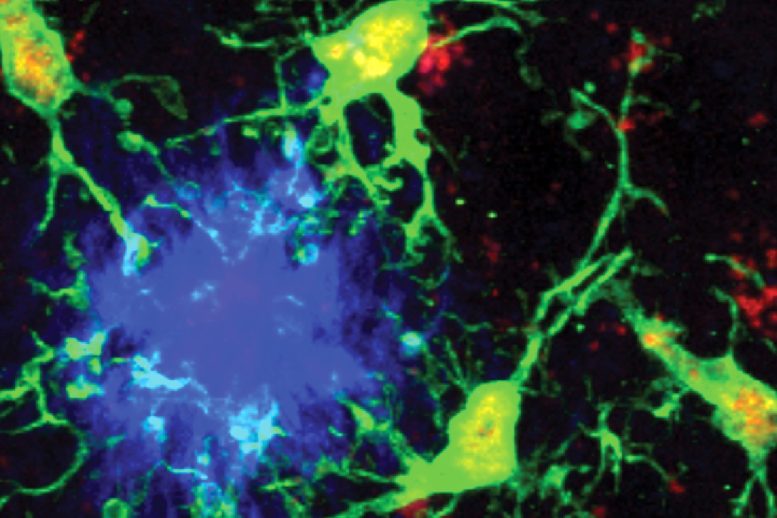
How PU.1 and CD28 Shape Immunity in the Brain
PU.1 is a protein that attaches to certain regions of DNA to direct how genetic information is used, ultimately controlling when genes turn on or off. CD28 is a co-stimulatory receptor that appears on T cells and helps strengthen and maintain an effective immune response.
Through experiments involving Alzheimer’s mouse models, human cells, and human brain tissue, the research team showed that lowering PU.1 levels encourages microglia to produce lymphoid immunoregulatory receptor proteins. Although these protective microglia make up a small portion of the total microglial population, they have wide-reaching effects, reducing inflammation throughout the brain and helping preserve memory and survival in mice. When CD28 was removed from this subset of microglia, inflammation increased and plaques grew more rapidly, revealing how essential CD28 is for their protective function.
Redefining Microglia: From Destroyers to Defenders
“Microglia are not simply destructive responders in Alzheimer’s disease—they can become the brain’s protectors,” said Anne Schaefer, MD, PhD, Professor in the Nash Family Department of Neuroscience at the Icahn School of Medicine, Co-Director of the Center for Glial Biology at The Friedman Brain Institute, Director of the Max Planck Institute for Biology of Ageing, and senior author of the paper.
“This finding extends our earlier observations on the remarkable plasticity of microglia states and their important roles in diverse brain functions. It also underscores the vital importance of international collaboration in advancing scientific progress.”
Shared Immune Logic Across Brain and Body
“It is remarkable to see that molecules long known to immunologists for their roles in B and T lymphocytes also regulate microglial activity,” added Alexander Tarakhovsky, MD, PhD, Dr. Plutarch Papamarkou Professor of Immunology, Virology, and Microbiology at The Rockefeller University and co-author of the paper.
“This discovery comes at a time when regulatory T cells have achieved major recognition as master regulators of immunity, highlighting a shared logic of immune regulation across cell types. It also paves the way for immunotherapeutic strategies for Alzheimer’s disease.”
The study builds on pioneering genetic work by Alison M. Goate, DPhil, Jean C. and James W. Crystal Professor of Genomics and Chair of the Department of Genetics and Genomic Sciences at the Icahn School of Medicine, founding director of the Ronald M. Loeb Center for Alzheimer’s Disease at Mount Sinai, and a senior co-author of the study, who identified a common variant in SPI1—the gene encoding PU.1—as being associated with reduced Alzheimer’s risk.
Toward Microglia-Targeted Immunotherapy
“These results provide a mechanistic explanation for why lower PU.1 levels are linked to reduced Alzheimer’s disease risk,” said Dr. Goate.
The discovery of the PU.1-CD28 axis establishes a molecular framework for understanding protective microglial states and highlights the potential of microglia-targeted immunotherapies to modify the course of Alzheimer’s disease.
Reference: “Lymphoid gene expression supports neuroprotective microglia function” by Pinar Ayata, Jessica M. Crowley, Matthew F. Challman, Vinaya Sahasrabuddhe, Maud Gratuze, Sebastian Werneburg, Diogo Ribeiro, Emma C. Hays, Violeta Durán-Laforet, Travis E. Faust, Philip Hwang, Francisco Mendes Lopes, Chrysa Nikopoulou, Sarah Buchholz, Robert E. Murphy, Taoyu Mei, Anna A. Pimenova, Carmen Romero-Molina, Francesca Garretti, Tulsi A. Patel, Claudia De Sanctis, Angie V. Ramirez Jimenez, Megan Crow, Felix D. Weiss, Jason D. Ulrich, Edoardo Marcora, John W. Murray, Felix Meissner, Andreas Beyer, Dan Hasson, John F. Crary, Dorothy P. Schafer, David M. Holtzman, Alison M. Goate, Alexander Tarakhovsky and Anne Schaefer, 5 November 2025, Nature.
DOI: 10.1038/s41586-025-09662-z
This research received support from a wide range of organizations, including the National Institutes of Health, European Research Council, Stavros Niarchos Foundation, Cure Alzheimer’s Fund, Freedom Together Foundation, Belfer Neurodegeneration Consortium Grant, Massachusetts Life Sciences Center, Robin Chemers Neustein Postdoctoral Fellowship Award, Alfred P. Sloan Foundation, Alzheimer’s Association, BrightFocus Foundation, National Multiple Sclerosis Society, and Clinical and Translational Science Awards.
Never miss a breakthrough: Join the SciTechDaily newsletter.
Follow us on Google and Google News.