Scientists have identified a key vulnerability in pancreatic cancer by targeting transcription-replication conflicts—cellular disruptions that cause genetic instability in tumors.
Their novel approach uses a drug called AOH1996, which slowed tumor growth in lab models and reduced tumors in human patients, showing promise where traditional treatments have failed. This breakthrough offers hope for fighting one of the deadliest cancers and paves the way for more precise, stress-targeted therapies, particularly in KRAS-mutated tumors.
New Target for Treating Pancreatic Cancer
Researchers at City of Hope, one of the nation’s leading cancer research and treatment centers, have identified a new molecular target for treating pancreatic cancer. The findings were published today (April 8) in the journal Gastroenterology.
Pancreatic ductal adenocarcinoma (PDAC) is among the deadliest cancers worldwide, largely because it resists most current treatments. With limited therapeutic options, about 90% of patients die within five years of diagnosis. Now, researchers have uncovered a promising new strategy that could turn a key genetic weakness in PDAC into a target for treatment.
Cracking Cancer’s Weak Spot
The study, led by Mustafa Raoof, M.D., M.S., assistant professor of surgery, cancer genetics, and epigenetics at City of Hope, focused on a cellular phenomenon known as transcription-replication conflicts (TRCs). These occur when the processes of gene expression and DNA duplication interfere with each other. In pancreatic cancer, this clash causes what’s known as replication stress, which leads to errors in DNA copying. These errors can help cancer grow and spread, making TRCs a potential weak point researchers aim to exploit.
“Transcription-replication conflicts are an important vulnerability of pancreatic cancer,” said Dr. Raoof, senior author of the new study. “Our study is the first to confirm proof of concept for whether exploiting this chink in cancer’s armor could provide an effective therapeutic target for patients.”
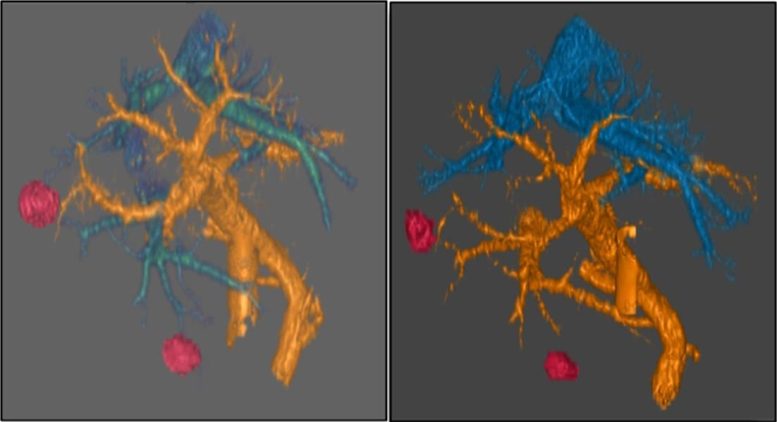
Testing a New Therapeutic Strategy
In an earlier study, Dr. Raoof and his colleagues had identified high levels of TRCs as a unique weakness in pancreatic cancers that are driven by a common gene mutation. Building upon this research, his team used an experimental drug developed at City of Hope called AOH1996 as a tool to target TRCs and measure clinical responses.
First, the laboratory tested AOH1996 on a mouse model for pancreatic cancer and on small, lab-grown versions of human organs called organoids. The scientists discovered that the drug slowed tumor growth, damaged tumor cells without harming healthy tissue and boosted mouse survival from a median of 14 days to three weeks.
Next, the team tested the approach on two patients whose pancreatic tumors had resisted earlier treatments (NCT05227326). The patients experienced up to a 49% shrinkage in their liver metastases after taking the pill twice a day for two months.
Effective Against KRAS-Driven Tumors
Overall, the experimental approach was most effective at killing cancer cells with high replication stress, a common phenomenon that occurs when the KRAS gene goes awry in 95% of patients with pancreatic cancer.
“While the KRAS mutation has suggested a strong therapeutic target, pinpointing it in human PDAC has been difficult until now,” said Dr. Raoof. “With inhibitors to mutant KRAS entering clinical trials, resistance is expected. It’s crucial for us to develop new approaches that target dependency on KRAS.”
Targeting TRCs enabled the scientists to pinpoint only pancreatic cancer cells that experienced high levels of replication stress.
“Transcription-replication conflicts are more prevalent in cancer cells than normal cells,” Dr. Raoof said. “Therapies that interfere with how cells manage their DNA during replication could open up new ways to treat cancer, offering hope for patients who have not benefited from other approaches.”
Cautious Optimism and Future Research
Though excited by the study’s early results, Dr. Raoof emphasized caution in interpreting its findings. Due to the trial’s small size, scientists will need to pursue larger clinical and biomarker discovery studies to realize the full potential of therapeutic targeting of TRCs.
Reference: “Therapeutic Targeting of Oncogene-induced Transcription-Replication Conflicts in Pancreatic Ductal Adenocarcinoma” by Shanna J. Smith, Fan Meng, Robert G. Lingeman, Caroline M. Li, Min Li, Galyah Boneh, Toni T. Seppälä, Thuy Phan, Haiqing Li, Richard A. Burkhart, Vishwas Parekh, Syed Rahmanuddin, Laleh G. Melstrom, Robert J. Hickey, Vincent Chung, Yilun Liu, Linda H. Malkas and Mustafa Raoof, 8 April 2025, Gastroenterology.
DOI: 10.1053/j.gastro.2025.02.038
The study was supported by grants from the National Comprehensive Cancer Network and the 2020 Pancreatic Cancer Action Network Career Development Award in Memory of Skip Viragh (20-20-RAOO) to the Raoof laboratory, as well as by the National Cancer Institute of the National Institutes of Health (P30CA033572) and NCCN Foundation.
A respected birthplace for biotech, City of Hope created the technology that led to the development of synthetic human insulin. City of Hope later contributed to the development of “smart” cancer drugs like Herceptin, Rituxan and Avastin.
City of Hope’s Linda Malkas, Ph.D., discovered and developed AOH1996, which is exclusively licensed to the biotechnology company RLL, LLC.
Last year City of Hope received a historic $150 million gift to fund pancreatic cancer research from two entrepreneur-philanthropists: A. Emmet Stephenson Jr. and his daughter, Tessa Stephenson Brand. The donation’s mission is to accelerate leading-edge research into effective pancreatic cancer treatments through scientific partnerships with the world’s top researchers, regardless of institutional affiliation.
Never miss a breakthrough: Join the SciTechDaily newsletter.
Follow us on Google and Google News.