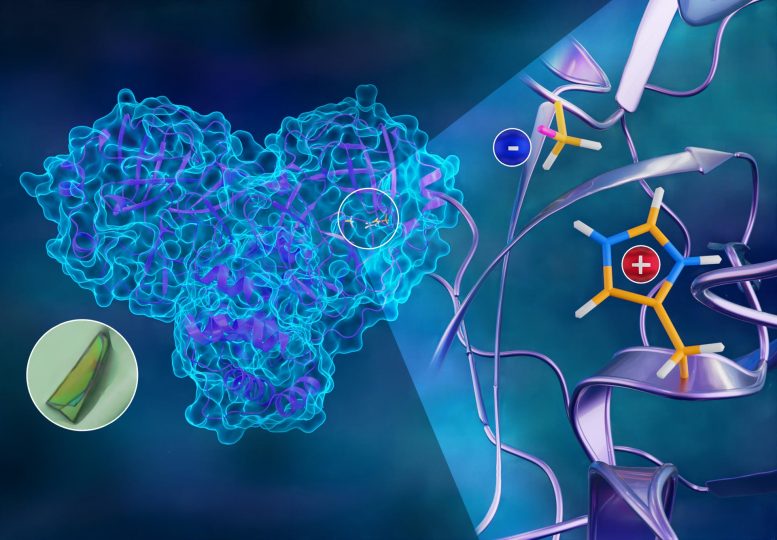
To better understand how the novel coronavirus behaves and how it can be stopped, scientists have completed a three-dimensional map that reveals the location of every atom in an enzyme molecule critical to SARS-CoV-2 reproduction.
Researchers at the Department of Energy’s Oak Ridge National Laboratory used neutron scattering to identify key information to improve the effectiveness of drug inhibitors designed to block the virus’s replication mechanism. The research was recently published in the Journal of Biological Chemistry.
The SARS-CoV-2 virus, which causes the COVID-19 disease, expresses long chains of proteins composed of approximately 1,900 amino acid residues. For the virus to reproduce, those chains have to be broken down and cut into smaller strands by an enzyme called the main protease. The active protease enzyme is formed from two identical protein molecules held together by hydrogen bonds. Developing a drug that inhibits or blocks the protease activity will prevent the virus from replicating and spreading to other cells in the body.
“This new information is exactly what is needed to design inhibitors with a higher degree of specificity, ensuring the inhibitor molecules are binding very tightly to their intended targets and disabling the protease,” said ORNL’s Andrey Kovalevsky, corresponding author.
Neutron experiments first revealed that the site containing the amino acids where the protein chains are cut is in an electrically charged reactive state and not in a resting or neutral state, contrary to previously held beliefs. Second, they mapped the location of each hydrogen atom in the places where inhibitors would bind to the protease enzyme, as well as the electrical charges of the associated amino acids. The experiments also charted the entire network of hydrogen bonds between the protein molecules that hold the enzyme together and enable it to initiate the chemical process of cutting the protein chains.
“Half of the atoms in proteins are hydrogen. Those atoms are key players in enzymatic function and are essential to how drugs bind,” Kovalevsky said. “If we don’t know where those hydrogens are and how the electrical charges are distributed inside the protein, we can’t design effective inhibitors for the enzyme.”
The team’s neutron study builds on previous research published in the journal Nature Communications, creating a complete atomic structure of the protease enzyme. The researchers have also made their data publicly available to the scientific community before both papers were published to accelerate solutions to the global pandemic.
Neutrons are ideal probes for studying biological structures because they are non-destructive and highly sensitive to light elements such as hydrogen. The neutron scattering experiments were performed at the High Flux Isotope Reactor and the Spallation Neutron Source at ORNL. The protein samples were synthesized in adjacent facilities at the Center for Structural Molecular Biology.
“This might be the quickest neutron structure of a protein ever produced. We started neutron experiments in May, and within five months, we obtained and published our results. That’s something that usually takes years,” said ORNL corresponding author Leighton Coates. “This work demonstrates what we can do at Oak Ridge. Everything was done here from start to finish. The proteins were expressed, purified, and crystallized, and all the data was collected and analyzed on-site — a completely vertically integrated approach.”
The team will now use the newly obtained information to investigate the binding properties of drug molecule candidates to produce improved COVID-19 therapeutics.
“Not only is this the first time anyone has obtained a neutron structure of a coronavirus protein, but it’s also the first time anyone has looked at this class of protease enzymes using neutrons,” said ORNL’s Daniel Kneller, the study’s first author. “It’s an outstanding example of neutron crystallography serving the community when it needs it the most.”
References:
“Unusual zwitterionic catalytic site of SARS-CoV-2 main protease revealed by neutron crystallography” by Daniel W. Kneller, Gwyndalyn Phillips, Kevin L Weiss, Swati Pant, Qiu Zhang, Hugh M O’Neill, Leighton Coates and Andrey Kovalevsky, 15 October 2020, Journal of Biological Chemistry.
DOI:
“Structural plasticity of SARS-CoV-2 3CL Mpro active site cavity revealed by room temperature X-ray crystallography” by Daniel W. Kneller, Gwyndalyn Phillips, Hugh M. O’Neill, Robert Jedrzejczak, Lucy Stols, Paul Langan, Andrzej Joachimiak, Leighton Coates and Andrey Kovalevsky, 24 June 2020, Nature Communications.
DOI: 10.1038/s41467-020-16954-7
In addition to Kneller, Coates, and Kovalevsky, the paper’s coauthors include Gwyndalyn Phillips, Kevin L. Weiss, Swati Pant, Qiu Zhang, and Hugh M. O’Neill.
COVID-19 research at ORNL is supported in part by the DOE Office of Science through the National Virtual Biotechnology Laboratory, a consortium of DOE national laboratories focused on response to COVID-19, with funding provided by the Coronavirus CARES Act.
HFIR and SNS are DOE Office of Science User Facilities.
Never miss a breakthrough: Join the SciTechDaily newsletter.
Follow us on Google and Google News.