
Alzheimer’s disease is a progressive brain disorder that affects memory, thinking, and behavior. It is the most common cause of dementia, a term used to describe a decline in cognitive function that affects daily life.
Researchers at the Indiana University School of Medicine are exploring the potential for reducing the risk of late-onset Alzheimer’s disease through the reduction of a gene variant in the brain’s immune cells.
The research, published in Alzheimer’s & Dementia: The Journal of the Alzheimer’s Association, was conducted by a research team led by Adrian Oblak, Ph.D., and Peter Bor-Chian Lin. Oblak is an assistant professor of radiology and imaging sciences, while Lin is a Ph.D. candidate in the Medical Neuroscience Graduate Program at Stark Neurosciences Research Institute.
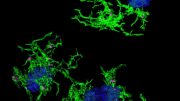
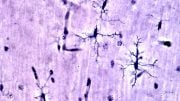
They focused their investigation on INPP5D, a microglia-specific gene that has been shown to increase the risk for developing late-onset Alzheimer’s disease. Microglia are the brain’s immune cells and there are multiple microglial genes associated with neurodegeneration.
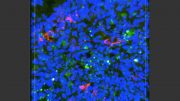
Oblak said the team’s previous data revealed that elevated levels of INPP5D in Alzheimer’s disease lab models resulted in increased plaque deposition. Knowing this, they aimed to understand how reducing the expression of INPP5D might regulate disease pathogenesis.
Using models in the lab, the researchers reduced the expression of the gene by at least 50 percent—called haplodeficiency—rather than completely knocking out the expression of the gene to mimic the treatment of pharmacological inhibitors targeting INPP5D as therapeutic strategies.
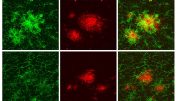
“INPP5D deficiency increases amyloid uptake and plaque engagement in microglia,” Oblak said. “Furthermore, inhibiting the gene regulates microglial functions and mitigates amyloid pathology that are likely mediated by TREM2-SYK signaling pathway activation.”
The gene deficiency also led to the preservation of cognitive function in the lab models. By reducing the expression of the gene in the brain, it created a less neurotoxic environment and improved the movement of microglia—which act as the first line of defense against viruses, toxic materials, and damaged neurons—to clear amyloid deposits and plaques.
“These findings suggest that mitigating the function of INPP5D can result in a protective response by diminishing disease risk and mitigating the effect of amyloid beta-induced pathogenesis,” Lin said.
Reference: “INPP5D deficiency attenuates amyloid pathology in a mouse model of Alzheimer’s disease” by Peter Bor-Chian Lin, Andy Po-Yi Tsai, Disha Soni, Audrey Lee-Gosselin, Miguel Moutinho, Shweta S. Puntambekar, Gary E. Landreth, Bruce T. Lamb and Adrian L. Oblak, 16 December 2022, Alzheimer’s & Dementia.
DOI: 10.1002/alz.12849


Be the first to comment on "How the Reduction of a Gene Variant Found in the Brain’s Immune Cells Could Diminish the Risk of Alzheimer’s Disease"