
The nanobody can also punch through tough brain cells.
The immune system uses proteins referred to as antibodies to detect and attack invading pathogens. Mini versions of antibodies, called nanobodies — natural compounds in the blood of animals such as llamas and sharks — are being researched to treat autoimmune diseases and cancer. Now, scientists from Johns Hopkins Medicine have helped create a nanobody that can penetrate the tough outer layer of brain cells and disentangle misshapen proteins that cause disorders such as Parkinson’s disease, Lewy body dementia, and other neurocognitive problems.
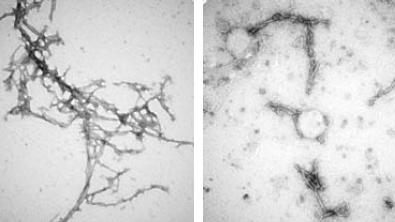
Researchers from Johns Hopkins Medicine, under the direction of Xiaobo Mao, Ph.D., and researchers from the University of Michigan, Ann Arbor, collaborated on the study, which was recently published in the journal Nature Communications. They set out to discover a new treatment method that could target the misshapen proteins known as alpha-synuclein, which have a tendency to cluster and impede the inner workings of brain cells. New research suggests that alpha-synuclein clumps can spread from the gut or nose to the brain, accelerating disease progression.
Theoretically, antibodies may be able to target clumping alpha-synuclein proteins, but pathogen-fighting compounds have difficulty penetrating the outer covering of brain cells. To get past the tough brain cell coatings, the researchers chose to employ nanobodies, which are tiny versions of antibodies.
Genetically Engineering Nanobodies for Brain Cell Stability
Traditionally, nanobodies produced outside of the cell may not perform the same function within the cell. As a result, the researchers had to strengthen the nanobodies in order for them to remain stable inside a brain cell. They achieved this by genetically engineering the nanobodies to purge them of the chemical bonds that normally degrade within a cell. Tests revealed that even without the bonds, the nanobody was still able to bind to misshapen alpha-synuclein and stay stable.

PFFNB2: A Nanobody with Potential
The team made seven, similar types of nanobodies, known as PFFNBs, that could bind to alpha-synuclein clumps. Of the nanobodies they created, one — PFFNB2 — did the best job of glomming onto alpha-synuclein clumps and not single molecules, or monomers of alpha-synuclein. Monomer versions of alpha-synuclein are not harmful and may have important functions in brain cells. The researchers also needed to determine if the PFFNB2 nanobody could remain stable and work inside brain cells. The team found that in live mouse brain cells and tissue, PFFNB2 was stable and showed a strong affinity to alpha-synuclein clumps rather than single alpha-synuclein monomers.
Additional tests in mice showed that the PFFNB2 nanobody cannot prevent alpha-synuclein from collecting into clumps, but it can disrupt and destabilize the structure of existing clumps.
“Strikingly, we induced PFFNB2 expression in the cortex, and it prevented alpha-synuclein clumps from spreading to the mouse brain’s cortex, the region responsible for cognition, movement, personality, and other high-order processes,” says Ramhari Kumbhar, Ph.D., the co-first author, a postdoctoral fellow at the Johns Hopkins University School of Medicine.
“The success of PFFNB2 in binding harmful alpha-synuclein clumps in increasingly complex environments indicates that the nanobody could be key to helping scientists study these diseases and eventually develop new treatments,” says Mao, associate professor of neurology.
Reference: “α-Synuclein fibril-specific nanobody reduces prion-like α-synuclein spreading in mice” by Yemima R. Butler, Yuqing Liu, Ramhari Kumbhar, Peiran Zhao, Kundlik Gadhave, Ning Wang, Yanmei Li, Xiaobo Mao and Wenjing Wang, 19 July 2022, Nature Communications.
DOI: 10.1038/s41467-022-31787-2
The study was funded by the University of Michigan, the National Institutes of Health, the Parkinson’s Foundation, and the Maryland Stem Cell Research Foundation.
Never miss a breakthrough: Join the SciTechDaily newsletter.
Follow us on Google and Google News.
1 Comment
Is this research complete? Do you need volunteers? Please send me the latest updates on your research.
Thanks,