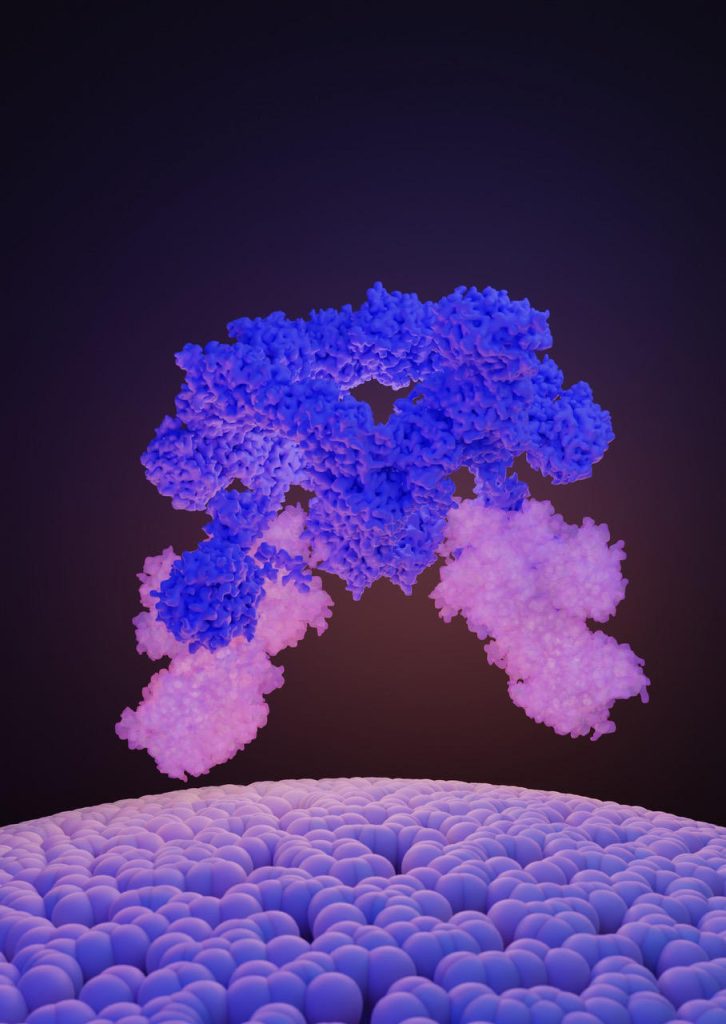
A UNIGE team has discovered the structure of a protein complex that regulates the activity of the major growth regulator.
The mTOR protein plays a central role in cell growth, proliferation, and survival. Its activity is affected by the availability of nutrients as well as various growth factors such as hormones. This protein has been linked to a number of diseases, including cancer, where its activity frequently increases.
A team from the University of Geneva (UNIGE), in collaboration with researchers from the Martin Luther University (MLU) of Halle-Wittenberg in Germany, and the recently inaugurated Dubochet Center for Imaging (UNIGE-UNIL-EPFL), has identified the structure of the SEA complex, an interdependent set of proteins responsible for controlling mTOR. The finding of this structure provides a better understanding of how cells perceive nutrient levels in order to control their growth. The study was recently published in the journal Nature.
From yeast to humans, the mTOR protein (mammalian target of rapamycin) is the central controller of cell growth. This protein reacts to environmental cues such as nutrients and hormones and controls several key cellular functions such as protein and lipid synthesis, energy production by mitochondria, and cell structure organization. mTOR activity disruptions are the root cause of many disorders, including diabetes, obesity, epilepsy, and several types of cancer.
Two Opposing Functions in the Same Complex
The laboratory of Robbie Loewith, Professor in the Department of Molecular and Cellular Biology at the UNIGE Faculty of Science and director of the National Center for Competence in Research in Chemical Biology, is interested in the regulation of mTOR, and in particular in the SEA complex, which is the direct sensor of nutrients and which controls the activity of mTOR. The SEA complex is composed of eight proteins. One part of the SEA complex (SEACIT) is involved in the inhibition of mTOR activity, while the other part (SEACAT) is involved in its activation.
In the absence of nutrients, the mTOR protein is blocked by the SEACIT subcomplex, and cell growth is thus prevented. In contrast, in the presence of nutrients, the SEACAT subcomplex is thought to inhibit the SEACIT subcomplex, which can no longer block the mTOR protein. The central controller can then exert its activating role in cell growth by, for example, stimulating the production of proteins and lipids. How SEACAT regulates SEACIT is still not understood.
Determining Structure To Understand the Function
To determine the interactions between the proteins of the SEA complex, and thus better understand how they work, the researchers set out to determine the structure of this complex. After biochemically separating the SEA complex from all of the other components in the cell, the scientists used the technologies of the Dubochet Center for Imaging of UNIGE, UNIL, and EPFL to obtain its molecular structure by cryo-electron microscopy (cryo-EM).
‘‘By freezing the samples very quickly at -180°C, cryo-EM allows to obtain the structure of the proteins in their original state, i.e. in their functional three-dimensional form,’’ explains Lucas Tafur, a researcher in the Department of Molecular and Cellular Biology and first author of the study.
SEACAT Is Necessary but Not Sufficient
The biochemical activities of the different components of the complex were then tested in the laboratory. Despite the SEACAT subcomplex being in an active form (as when in the presence of nutrients), the researchers observed that the SEACIT subcomplex is still active and capable of blocking mTOR.
‘‘This result is very unexpected since SEACAT has long been described as the direct inhibitor of SEACIT. We, therefore, expected SEACIT to be inactive in the presence of active SEACAT. Our results show that SEACAT acts more as a scaffold for the recruitment of other regulatory proteins and that its presence is, therefore, necessary but not sufficient for the inhibition of SEACIT,’’ explains Robbie Loewith, the last author of the study.
Obtaining the structure of the SEA complex has allowed highlighting missing links in the mTOR regulatory cascade. ‘‘Of course, we now need to identify the as-yet unknown partners that associate with this complex. These new factors could prove to be therapeutic targets for tumors where mTOR activity is exacerbated,’’ concludes Lucas Tafur.
Reference: “Cryo-EM structure of the SEA complex” by Lucas Tafur, Kerstin Hinterndorfer, Caroline Gabus, Chiara Lamanna, Ariane Bergmann, Yashar Sadian, Farzad Hamdi, Fotis L. Kyrilis, Panagiotis L. Kastritis and Robbie Loewith, 26 October 2022, Nature.
DOI: 10.1038/s41586-022-05370-0
Never miss a breakthrough: Join the SciTechDaily newsletter.
Follow us on Google and Google News.