Salk scientists discover how microprotein PIGBOS could be a target for cancer.
As the tools to study biology improve, researchers are beginning to uncover details in microproteins, small components that appear to be key to some cellular processes, including those involved with cancer. Proteins are made up of chains of linked amino acids and the average human protein contains around 300 amino acids. Meanwhile, microproteins have fewer than 100 amino acids.
One such microprotein is the 54-amino acid microprotein called PIGBOS, which Salk scientists recently showed contributes to mitigating cell stress. The work, published on October 25, 2019, in the journal Nature Communications, indicates that PIGBOS could be a target for human disease.
“This study is exciting because cell stress is important in a number of different diseases, including cancer and neurodegeneration,” says Salk Professor Alan Saghatelian, co-corresponding author of the study. “By understanding the mechanisms behind these diseases, we think we’ll have a better shot at treating them.”
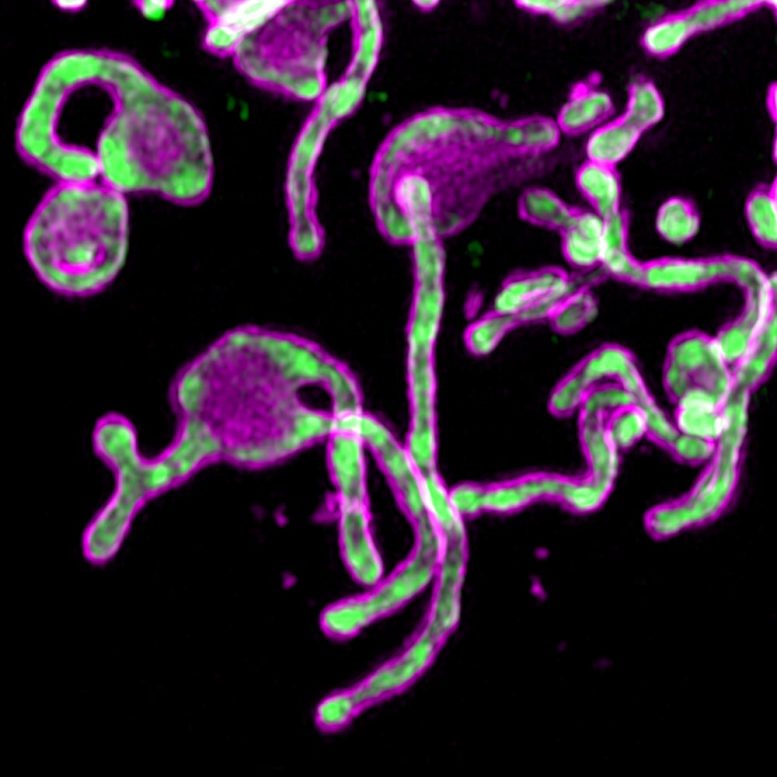
As the tools to study biology improve, researchers are beginning to uncover details into microproteins, small components that appear to be key to some cellular processes. The lab of Salk Professor Alan Saghatelian, along with Uri Manor, director of the Waitt Advanced Biophotonics Core Facility, recently showed that the 54-amino acid microprotein PIGBOS contributes to mitigating cell stress. The work, published in the journal Nature Communications, indicates that PIGBOS could be a target for human disease. Credit: Salk Institute
The study began when Salk postdoctoral researcher and first author Qian Chu detected PIGBOS in mitochondria, small organelles that power important cell functions. Chu wondered what PIGBOS’ role could be. He knew it wouldn’t be easy to find the answer. Researchers had previously noted the gene that could code for PIGBOS, but no one knew where to find the protein or what it did in cells.
That’s when the team reached out to co-corresponding author Uri Manor, director of the Waitt Advanced Biophotonics Core Facility at Salk. Manor’s team uses tools like fluorescent protein tags to locate proteins and see what they are doing in cells.
“Only now do we really have the sophisticated tools to probe interactions between proteins and see how they work and how they are regulated,” says Manor.
But Manor ran into a roadblock when he tried to attach a common tag, called green fluorescent protein (GFP), to PIGBOS. The microprotein was just too small relative to the size of GFP. Manor’s team solved this problem by trying a less common approach called split GFP, where they fused just a small part of GFP, called a beta strand, to PIGBOS.
At last, the researchers could see PIGBOS and study how it interacted with other proteins. As they mapped PIGBOS’ location, they realized that it sits on the outer membrane of the mitochondria, poised to make contact with proteins on other organelles. They were surprised to see PIGBOS interacting with a protein called CLCC1, which is part of an organelle called the endoplasmic reticulum (ER).
“PIGBOS is like a connection to link mitochondria and ER together,” says Chu. “We hadn’t seen that before in microproteins—and it’s rare in just normal proteins.”

The researchers found that PIGBOS actually communicates with CLCC1 to regulate stress in the ER. Without PIGBOS, the ER is more likely to experience stress, which leads to a chain of events where the cell tries to clear out harmful misshapen proteins (called the unfolded protein response). If the cell fails to dispose of these proteins, it will initiate a self-destruct sequence and die.
The scientists were not expecting to see a role for a mitochondrial protein in the unfolded protein response. This new understanding of PIGBOS opens the door to future therapies that can target cell stress.
“Going forward, we might consider how PIGBOS is involved in diseases like cancer,” says Chu. “In cancer patients, the ER is more stressed than in a normal person, so ER stress regulation could be a good target.”
The researchers are interested in studying the roles of other mitochondrial proteins in ER stress, and in exploring how PIGBOS works in an animal model. The team is also forging ahead in characterizing the vast library of microproteins that may be crucial in cell biology.
“Microproteins represent a fledgling field,” says Saghatelian. “But I think this work has really impacted our understanding of the impact that microproteins can have on biochemistry and cell biology.”
Manor adds, “PIGBOS represents one of a limited set of microproteins that anyone has gone through the effort to characterize. And lo and behold it actually has a very important role.”
Reference: “Regulation of the ER stress response by a mitochondrial microprotein” by Qian Chu, Thomas F. Martinez, Sammy Weiser Novak, Cynthia J. Donaldson, Dan Tan, Joan M. Vaughan, Tina Chang, Jolene K. Diedrich, Leo Andrade, Andrew Kim, Tong Zhang, Uri Manor and Alan Saghatelian, 25 October 2019, Nature Communications.
DOI: 10.1038/s41467-019-12816-z
Other authors of the study included Thomas F. Martinez, Sammy Weiser Novak, Cynthia J. Donaldson, Dan Tan, Joan M. Vaughan, Tina Chang, Jolene K. Diedrich, Leo Andrade, Andrew Kim and Tong Zhang of Salk.
The research was supported by the National Institutes of Health (R01GM102491, P30 014195, NS072031, P30 CA014195), the Leona M. and Harry B. Helmsley Charitable Trust grant, Dr. Frederick Paulsen Chair/Ferring Pharmaceuticals, the George E. Hewitt Foundation for Medical Research, the Anderson Foundation, the Waitt Foundation, and a National Institutes of Health F32 fellowship (GM123685) and Pioneer Fellowship.
Never miss a breakthrough: Join the SciTechDaily newsletter.
Follow us on Google and Google News.
1 Comment
Is there a picture anywhere of what Airplane air might look like? Air compressed in a small area like on overnight flight might contain – many interesting visuals, actually seeing such things as viruses floating around????