
Curiosity’s findings of elevated C12 on Mars hint at possible past life.
As far as we know, carbon is critical to life. So anytime we detect a strong carbon signature somewhere like Mars, it could mean biological activity.
Does a strong carbon signal in Martian rocks indicate biological processes of some type?
Any strong carbon signal is intriguing when you’re hunting for life. It’s a common element in all the forms of life we know of. But there are different types of carbon, and carbon can become concentrated in the environment for other reasons. It doesn’t automatically mean life is involved in carbon signatures.
Understanding Carbon Isotopes and Their Significance
Carbon atoms always have six protons, but the neutron count can vary. Carbon atoms with different numbers of neutrons are called isotopes. Three carbon isotopes occur naturally: C12 and C13, which are stable, and C14, a radionuclide. C12 has six neutrons, C13 has seven neutrons, and C14 has eight neutrons.
When it comes to carbon isotopes, life prefers C12. They use it in photosynthesis or to metabolize food. The reason is relatively simple. C12 has one fewer neutron than C13, which means that when it bonds with other atoms into molecules, it makes fewer connections than C13 does in the same situation. Life is essentially lazy, and it will always seek the easiest way to do things. C12 is easier to use because it forms fewer bonds than C13. It’s easier to get at than C13, and life never takes the hard way when an easier way is available.
Curiosity Rover’s Findings in Gale Crater
The Curiosity rover is hard at work in Mars’ Gale Crater, searching for signs of life. It drills into rock, extracts a pulverized sample, and places it into its onboard chemistry laboratory. Curiosity’s lab is called SAM which stands for Sample Analysis at Mars. Inside SAM, the rover uses pyrolysis to bake the sample and convert the carbon in the rock into methane. The pyrolysis is done in a flow of inert helium to prevent any contamination in the process. Then it probes the gas with an instrument named the Tunable Laser Spectrometer to find out what carbon isotopes are in the methane.

The team behind Curiosity’s SAM looked at 24 rock samples with this process and recently discovered something noteworthy. Six of the samples showed elevated ratios of C12 to C13. Compared to an Earth-based reference standard for C12/C13 ratios, the samples from these six sites contained greater than 70 parts per thousand more C12. On Earth, 98.93% of the carbon is C12 Earth, and C13 forms the remaining 1.07%.
A new study published in the Proceedings of the National Academy of Sciences (PNAS) presented the findings. Its title is “Depleted carbon isotope compositions observed at Gale crater, Mars.” The lead author is Christopher House, a Curiosity scientist at Penn State University.
Exploring Non-Biological Explanations for Carbon Signatures
It’s an exciting finding, and if these results were obtained on Earth, they would signal that a biological process produced the abundance of C12.
On ancient Earth, surface bacteria produced methane as a byproduct. They’re called methanogens, and they’re prokaryotes from the Archaea domain. Methanogens are still present today on Earth, in anoxic wetlands, in the digestive tracts of ruminants, and extreme environments like hot springs.
These bacteria produce methane that enters the atmosphere, interacting with ultraviolet light. Those interactions produce more complex molecules that rained down onto the Earth’s surface. They’re preserved in Earth rocks, along with their carbon signatures. The same thing might have happened on Mars, and if it did, it could account for Curiosity’s findings.
But this is Mars. If the history of searching for life on Mars tells us anything, it’s not to get ahead of ourselves.
“We’re finding things on Mars that are tantalizingly interesting, but we would really need more evidence to say we’ve identified life,” said Paul Mahaffy, former principal investigator for Curiosity’s Sample Analysis at Mars lab. “So we’re looking at what else could have caused the carbon signature we’re seeing, if not life.”

In their paper, the authors write, “There are multiple plausible explanations for the anomalously depleted 13C observed in evolved methane, but no single explanation can be accepted without further research.”
Considering Mars’ Unique Environment and Processes
One of the difficulties in understanding carbon signatures like this one is our so-called Earth bias. Most of what scientists know about atmospheric chemistry and related things is based on Earth. So when it comes to this newly-detected carbon signature on Mars, scientists can find it challenging to keep their minds open to new possibilities that may not exist on Mars. The history of the search for life on Mars tells us this.
“The hardest thing is letting go of Earth and letting go of that bias that we have and really trying to get into the fundamentals of the chemistry, physics and environmental processes on Mars,” said Goddard astrobiologist Jennifer L. Eigenbrode, who participated in the carbon study. Previously, Eigenbrode led an international team of Curiosity scientists in the detection of myriad organic molecules — ones that contain carbon — on the Martian surface.
“We need to open our minds and think outside the box,” Eigenbrode said, “and that’s what this paper does.”
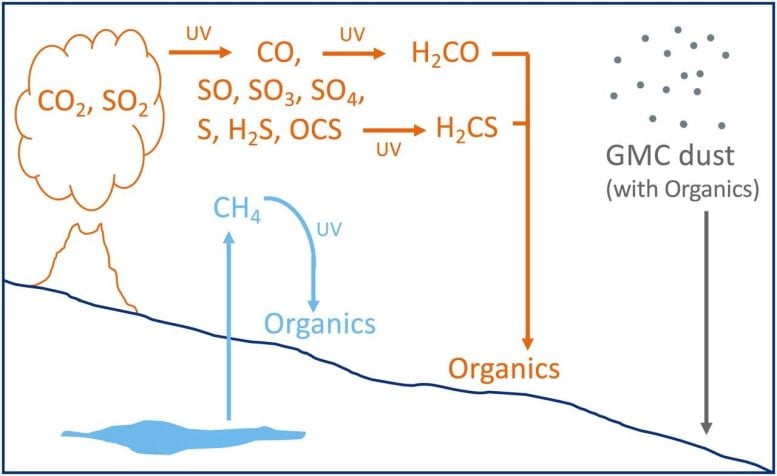
The researchers point out two non-biological explanations for the unusual carbon signature in their paper. One involves molecular clouds.
Non-Biological Hypotheses: Molecular Clouds and UV Light
The molecular cloud hypothesis states that our Solar System passed through a molecular cloud hundreds of millions of years ago. That is a rare event, but it happens about once every 100 million years, so scientists can’t discount it. Molecular clouds are primarily molecular hydrogen, but one may have been rich in the type of lighter carbon detected by Curiosity in Gale Crater. The cloud would’ve caused Mars to cool dramatically, causing glaciation in this scenario. The cooling and glaciation would’ve prevented the lighter carbon in the molecular clouds from mixing with Mars’ other carbon, creating deposits of elevated C12. The paper states that “Glacial melt during the glacial period and ice retreat after should leave the interstellar dust particles on the glacial geomorphological surface.”
The hypothesis fits since Curiosity found some of the elevated C12 levels at the tops of ridges—such as the top of Vera Rubin Ridge—and other high points in Gale Crater. The samples were gathered from “… a variety of lithologies (mudstone, sand, and sandstone) and are temporally spread throughout the mission operations to date,” the paper states. Still, the molecular cloud hypothesis is an unlikely chain of events.

The other non-biological hypothesis involves ultraviolet light. Mars’ atmosphere is over 95% carbon dioxide, and in this scenario, UV light would’ve interacted with carbon dioxide gas in Mars’ atmosphere producing new carbon-containing molecules. The molecules would’ve rained down on Mars’ surface and become part of the rock there. This hypothesis is similar to how methanogens indirectly produce C12 on Earth, but it’s entirely abiotic.
The Need for Further Research and Caution
“All three explanations fit the data,” said lead author Christopher House. “We simply need more data to rule them in or out.”
“On Earth, processes that would produce the carbon signal we’re detecting on Mars are biological,” House added. “We have to understand whether the same explanation works for Mars or if there are other explanations because Mars is very different.”
Almost half of the Curiosity samples had unexpectedly elevated levels of C12. They’re not only higher than Earth’s ratio; they’re higher than scientists have found in Martian meteorites and the Martian atmosphere. The samples came from five locations in Gale Crater, and all the locations had one thing in common: they have ancient, well-preserved surfaces.
As Paul Mahaffy said, the findings are “tantalizingly interesting.” But scientists are still learning about Mars’ carbon cycle, and there’s a lot we’re still ignorant about. It’s tempting to make assumptions about Mars’ carbon cycle based on Earth’s carbon cycle. But carbon may cycle through Mars in ways we haven’t even guessed at yet. Whether or not this carbon signature ends up being a signal for life or not, it’s still valuable knowledge when it comes to understanding Mars’ carbon signature.
“Defining the carbon cycle on Mars is absolutely key to trying to understand how life could fit into that cycle,” said Andrew Steele, a Curiosity scientist based at the Carnegie Institution for Science in Washington, D.C. “We have done that really successfully on Earth, but we are just beginning to define that cycle for Mars.”
But it isn’t easy to draw conclusions about Mars based on Earth’s carbon cycle. Steele made that clear when he said, “There’s a huge chunk of the carbon cycle on Earth that involves life, and because of life, there is a chunk of the carbon cycle on Earth we can’t understand because everywhere we look, there is life.”

Moving Forward: The Role of Curiosity and Perseverance Rovers
Curiosity is still working on Mars and will be for a while yet. The meaning of these samples, along with a better understanding of Mars’ carbon cycle, lies ahead. Curiosity will sample more rock to measure carbon isotope concentrations. It’ll sample rock from other well-preserved ancient surfaces to see if results are similar to these. Ideally, it would encounter another methane plume and sample it, but those events are unpredictable, and there’s no way to prepare for one.
Either way, these results will help inform Perseverance’s sample collecting at Jezero Crater. Perseverance may confirm similar carbon signals and even determine if they’re biological or not.
Perseverance is also gathering samples for return to Earth. Scientists will study those samples more effectively than the rover’s onboard lab can, so who knows what we’ll learn.
Ancient life on Mars is a tantalizing prospect, but for now, at least, it’s uncertain.
Adapted from an article originally published on Universe Today.
For more on this research, see:
- Newly Discovered Carbon on Mars: Origin May Be Biologically Produced Methane
- NASA’s Curiosity Rover Measures Intriguing Carbon Signature on Mars – Possible Indication of Biological Activity
Reference: “Depleted carbon isotope compositions observed at Gale crater, Mars” by Christopher H. House, Gregory M. Wong, Christopher R. Webster, Gregory J. Flesch, Heather B. Franz, Jennifer C. Stern, Alex Pavlov, Sushil K. Atreya, Jennifer L. Eigenbrode, Alexis Gilbert, Amy E. Hofmann, Maëva Millan, Andrew Steele, Daniel P. Glavin, Charles A. Malespin and Paul R. Mahaffy, 17 January 2022, Proceedings of the National Academy of Sciences.
DOI: 10.1073/pnas.2115651119
Never miss a breakthrough: Join the SciTechDaily newsletter.
Follow us on Google and Google News.
19 Comments
The part about C12 forming fewer bonds than other isotopes of carbon is so wrong it brings into doubt the accuracy of everything else written in the article.
Okay c12 🤔 h20 Mars intime 🍎🍊🍌🍇🧊 .4real only time …. 🍃🍀🍃🍀🦠🐌🐛 nasal don’t want earth to know there’s life on Mars 🕸🕷🕸🕷🕸🕷🦂🕸🕷🦗🕸🕷🦗🐜🐝🕸🐝🐜🕸
nasa not nasal my typo 🍕🍕🍕🍕🥞🥞🥞🥞
The C-13 chemistry description in the 3rd full paragraph is frighteningly wrong. C-13’s extra neutron does not create extra chemical bonds. Earth-based life’s preference for C-12 over C-13 is based on simple kinetics: photosynthetic and carbonate-forming processes gradually winnow out the slightly heavier isotope, leaving a preponderance of C-12.
http://www.madsci.org/posts/archives/Jun2003/1055532737.Bc.r.html#:~:text=The%20reason%20we%20see%20a,the%20more%20energetic%2012C%20atoms.
Neutrons do not engage in chemical reactions.
Enzymes can be selective.
The only time I can think of neutrons having any impact is when they decay into a proton, electron, and electron antineutrino.
Then the atom has changed proton number which will change bonding behavior. But that’s really it.
If you would take time to look a NASA’S picture of Greenheugh Pediment you would see extant and/or fossil microbes (?Archaea) growing and preserved in/on the Martian landscape. Whether NASA want’s to acknowledge that fact is up to them but it can be shown that MARS is covered with microbes and probably has been since early in its history. Sorry I can’t share pictures with you as this sight does not allow it.
Sorry for any typo’s.
Babu G. Ranganathan*
(B.A. Bible/Biology)
POSSIBLY MILLIONS OF TONS OF VOLCANIC EARTH SOIL REACHED MARS (Newsweek)
A Newsweek article of September 21, 1998, p.12 mentions the high possibility of Earth life on Mars because of millions of tons of Earth soil ejected into space from ancient volcanic explosions. “We think there’s about 7 million tons of earth soil sitting on Mars”, says USC scientist Kenneth Nealson. “You have to consider the possibility that if we find life on Mars, it could have come from the Earth” [Weingarten, T., Newsweek, September 21, 1998, p.12]. This may also explain why life forms may exist on Venus, again because they originated from Earth.
In the Earth’s past there was powerful volcanic activity which could have easily spewed dirt and rocks containing microbes and life into outer space which not only could have eventually reached Mars but also ended up traveling in orbit through space that we now know as meteors, comets, and asteroids. This would mean life forms found in meteorites originated from Earth in the first place.
Secular scientists have a different explanation from creationist scientists on the volcanic eruptions of the Earth’s past. Creation scientists believe, as Genesis teaches, that as the fountains of the deep were opened to release water for the world-wide flood the force of the eruptions could have indeed spewed great amounts of earth soil into space.
Life could not have evolved. A partially evolved cell would quickly disintegrate under the effects of random forces of the environment, especially without the protection of a complete and fully functioning cell membrane. A partially evolved cell cannot wait millions of years for chance to make it complete and living! In fact, it couldn’t have even reached the partially evolved state.
Having the right conditions and raw material for life do not mean that life can originate or arise by chance. Stanley Miller, in his famous experiment in 1953, showed that individual amino acids (the building blocks of life) could come into existence by chance. But, it’s not enough just to have amino acids. The various amino acids that make-up life must link together in a precise sequence, just like the letters in a sentence, to form functioning protein molecules. If they’re not in the right sequence the protein molecules won’t work. It has never been shown that various amino acids can bind together into a sequence by chance to form protein molecules. Even the simplest cell is made up of many millions of various protein molecules.
The probability of just an average size protein molecule arising by chance is 10 to the 65th power. Mathematicians have said any event in the universe with odds of 10 to 50th power or greater is impossible! The late great British scientist Sir Frederick Hoyle calculated that the odds of even the simplest cell coming into existence by chance is 10 to the 40,000th power! How large is this? Consider that the total number of atoms in our universe is 10 to the 82nd power.
Also, what many don’t realize is that Miller had a laboratory apparatus that shielded and protected the individual amino acids the moment they were formed, otherwise the amino acids would have quickly disintegrated and been destroyed in the mix of random energy and forces involved in Miller’s experiment.
Miller’s experiment produced equally both left-handed and right-handed amino acids, but all living things strictly require only left-handed amino acids. If a right-handed amino acid gets into the chain the protein won’t work.
There is no innate chemical tendency for the various amino acids to bond with one another in a sequence. Any one amino acid can just as easily bond with any other. The only reason at all for why the various amino acids bond with one another in a precise sequence in the cells of our bodies is because they’re directed to do so by an already existing sequence of molecules found in our genetic code.
Of course, once you have a complete and living cell then the genetic code and biological machinery exist to direct the formation of more cells, but how could life or the cell have naturally originated when no directing code and mechanisms existed in nature? Read my Internet article: HOW FORENSIC SCIENCE REFUTES ATHEISM.
Visit my newest Internet site: THE SCIENCE SUPPORTING CREATION
Author of popular Internet article, TRADITIONAL DOCTRINE OF HELL EVOLVED FROM GREEK ROOTS
* I have had the privilege of being recognized in the 24th edition of Marquis “Who’s Who In The East” for my writings on religion and science, and I have given successful lectures (with question and answer time afterwards) defending creation from science before evolutionist science faculty and students at various colleges and universities.
All these missions to find life on mars are a waste of time and money. We spend billions on these missions to find life and when we find evidence the same scientists that wanted the missions, say it is probably not life then find numerous things that could explain it’s non life origin. So what is the point of these missions? It would be way cheaper and more informative about the is there life on other planets, to simply look for molecules that cannot be created by nature, either in the atmosphere of planets or the soil. Looking for isotopes that could be created from numerous non organic origins is a waste of time and money.
The nuclear composition has nothing to do with the chemistry of life. Six electrons orbit the C12, C13, and C14.
Sorry that was me. I couldn’t find a bathroom so I just popped a squat behind a rock.
Life is lazy? This comes across as so dismissive and condescending of something we still don’t fully understand. I’m no expert but I have seen so many ways in which life is amazingly efficient, which I think is a better word than lazy.
We can get rocks from Mars.
Please, please get a real scientist to look at this first. The part about C12 and C13 is horribly wrong. So wrong it calls into question the entire article. This is wrong at a high school science level.
I don’t believe that the chemical behavior of a particular atom can change with Neutron (isotope) number. The chemical behavior is determined purely by the Proton count. The only thing that an isotope can add to a molecule is weight, hence heavy water. The deuterium isotope of Hydrogen makes water weigh a bit more, but has no effect on it’s chemical reactivity. There might be a bit of slowing down of a reaction because of the increased weight of the higher neutron count isotopes. But in general Heavy water behaves chemically the same as regular water. 10% of our bodies are made from heavy water. 10% of the water on the Earth is Heavy Water.
This is just… hilarious.
The number of bonds a carbon atom can make has almost nothing to do with neutrons. Barring an odd edge case I may not be familiar with, all carbon isotopes form the same number of bonds (4).
C12? 4 bonds.
C13? 4 bonds.
C14, 4 bonds until decays into nitrogen-14. So, I mean, neutrons can impact bonding but only through nuclear reactions transmuting nuclei into something with different bonding behavior.
If Mr. Gough’s assertion were true, C13 NMR wouldn’t be useful because C12 isn’t NMR active. Also, radiocarbon dating wouldn’t work, either because it relies on the fact that the C14 concentration in living bodies is the same as the C14 concentration in the atmosphere.
One thing is certain. If the strong isotopic results are to be from life on Mars billions of years ago, there must be carbon residues of these microbes in the sediments…fossil menthaogens or cyanobacteria. So far, zero carbon residues have been found anywhere on Mars.
I will try to attach a couple of pictures but I am not sure it will work. Sorry, this site is not going to allow me to add attachments. If interested, please drop me an email.