Ni/CaNH catalyst boosts ammonia-splitting efficiency, accelerating progress toward cleaner hydrogen energy.
Ammonia, a carbon-free resource can be split into nitrogen and hydrogen gas with the help of metal catalysts like Nickel (Ni). However, these reactions often require very high operating temperatures. Now scientists from Tokyo Institute of Technology (Tokyo Tech) have developed a highly efficient calcium imide (CaNH)-supported Ni catalyst that can decompose ammonia at temperatures 100°C lower than what conventional Ni catalysts require. This promising new catalyst can get us closer to sustainably producing hydrogen fuel.
The current global climate emergency and our rapidly receding energy resources have people looking out for cleaner alternatives like hydrogen fuel. When burnt in the presence of oxygen, hydrogen gas generates huge amounts of energy but none of the harmful greenhouse gases, unlike fossil fuels. Unfortunately, most of the hydrogen fuel produced today comes from natural gas or fossil fuels, which ultimately increases its carbon footprint.
Ammonia (NH3), a carbon-neutral hydrogen compound has recently garnered a lot of attention owing to its high energy density and high hydrogen storage capacity. It can be decomposed to release nitrogen and hydrogen gases. Ammonia can be easily liquified, stored, and transported, and converted into hydrogen fuel when required. However, the production of hydrogen from ammonia is a slow reaction with very high energy demands. To speed up production, metal catalysts are often used, which help reduce the overall energy consumption during hydrogen production as well.
Recent studies have found that Nickel (Ni) is a promising catalyst for splitting ammonia. Ammonia gets adsorbed on the surface of Ni catalysts, following which the bonds between nitrogen and hydrogen in ammonia are broken and they are released as individual gases. However, obtaining a good conversion of ammonia using a Ni catalyst often involves very high operating temperatures.
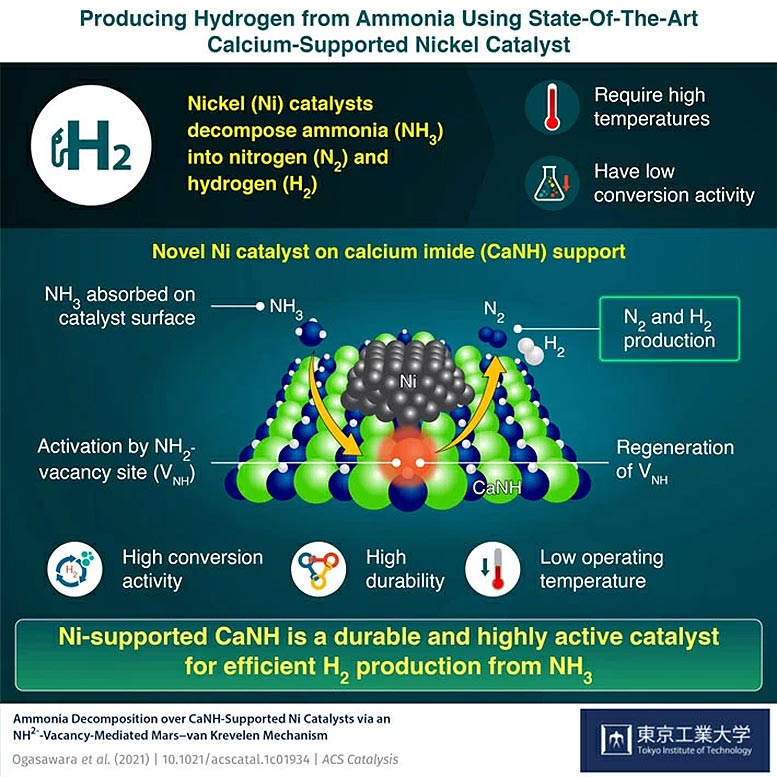
In a recent study published in ACS Catalysis, a team of researchers from Tokyo Tech led by Associate Professor Masaaki Kitano described a solution to overcome the issues faced by Ni-based catalysts. They developed a state-of-the-art calcium imide (CaNH)-supported Ni-catalyst that can achieve good ammonia conversion at lower operating temperatures. Dr. Kitano explains, “Our aim was to develop a highly active catalyst that would be energy efficient. Our addition of the metal imide to the catalyst system not only improved its catalytic activity but also helped us unravel the elusive working mechanism of such systems.”
Mars-van Krevelen Pathway
The team discovered that the presence of CaNH resulted in the formation of NH2- vacancies (VNH) on the surface of the catalyst. These active species resulted in the improved catalytic performance of the Ni/CaNH at reaction temperatures that were 100°C (180°F) lower than those necessary for the functioning of Ni-based catalysts. The researchers also developed computational models and conducted isotope-labeling to understand what is happening on the catalyst surface. The calculations proposed a Mars-van Krevelen mechanism that involved adsorption of ammonia onto the CaNH surface, its activation at the NH2- vacancy sites, formation of nitrogen and hydrogen gas, and finally regeneration of vacancy sites promoted by Ni nanoparticles.
The highly active and durable Ni/CaNH catalyst can be successfully deployed for the generation of hydrogen gas from ammonia. Also, the insight into the mechanism of catalysis provided by this study can be utilized to develop a new generation of catalysts. “As the whole world is working together to build a sustainable future, our research is aimed at resolving the hiccups faced on our way to a cleaner hydrogen fuel economy,” concludes Dr. Kitano.
This is a ray of hope for the world’s low carbon-emission mission!
Reference: “Ammonia Decomposition over CaNH-Supported Ni Catalysts via an NH2–-Vacancy-Mediated Mars–van Krevelen Mechanism” by Kiya Ogasawara, Takuya Nakao, Kazuhisa Kishida, Tian-Nan Ye, Yangfan Lu, Hitoshi Abe, Yasuhiro Niwa, Masato Sasase, Masaaki Kitano and Hideo Hosono, 19 August 2021, ACS Catalysis.
DOI: 10.1021/acscatal.1c01934
Never miss a breakthrough: Join the SciTechDaily newsletter.
Follow us on Google and Google News.
2 Comments
So where do we get the ammonia to make hydrogen from. I know, lets make it from Nitrogen & Hydrogen in the Haber Bosch process which requires considerable energy. Converting Ammonia back to H&N also requires energy, so what’s the point?
So when did ammonia become a carbon free resource, it’s an energy carrier just like hydrogen is and making both is usually carbon intensive. Anyway ammonia can be combusted directly to release N & H2O and was used as a temporary transport fuel during WW1.
For those interested in ammonia as a potential fuel, the article on New Atlas “green ammonia: the the rocky pathway to a clean new fuel” gives a far clearer picture of the pluses and minuses of ammonia vs hydrogen with many energy in & out numbers from CSIRO in Australia.
When ammonia is used as a fuel, it only returns about 21% of the input energy needed to make it. If the ammonia is turned back to hydrogen, then 19% of the original energy is available. It is also possible for ammonia to return 50% of the input energy when used with expensive SOFC technology. There is more, green vs blue vs brown ammonia same ideas as with hydrogen. The energy density of ammonia is about half that of diesel but 20* that of Li-ion batteries while being 30* less expensive to store than hydrogen. This would make ammonia a possible fuel for shipping if diesel is banned.