
New blueprint for affordable, sustainable ‘flow batteries’ developed at Berkeley Lab for renewable energy could accelerate an electrical grid powered by the sun and wind.
How do you store renewable energy so it’s there when you need it, even when the sun isn’t shining or the wind isn’t blowing? Giant batteries designed for the electrical grid — called flow batteries, which store electricity in tanks of liquid electrolyte — could be the answer, but so far utilities have yet to find a cost-effective battery that can reliably power thousands of homes throughout a lifecycle of 10 to 20 years.
Now, a battery membrane technology developed by researchers at the U.S. Department of Energy’s Lawrence Berkeley National Laboratory (Berkeley Lab) may point to a solution.
As reported in the journal of Joule, the researchers developed a versatile yet affordable battery membrane — from a class of polymers known as AquaPIMs. This class of polymers makes long-lasting and low-cost grid batteries possible based solely on readily available materials such as zinc, iron, and water. The team also developed a simple model showing how different battery membranes impact the lifetime of the battery, which is expected to accelerate early stage R&D for flow-battery technologies, particularly in the search for a suitable membrane for different battery chemistries.
“Our AquaPIM membrane technology is well-positioned to accelerate the path to market for flow batteries that use scalable, low-cost, water-based chemistries,” said Brett Helms, a principal investigator in the Joint Center for Energy Storage Research (JCESR) and staff scientist at Berkeley Lab’s Molecular Foundry who led the study. “By using our technology and accompanying empirical models for battery performance and lifetime, other researchers will be able to quickly evaluate the readiness of each component that goes into the battery, from the membrane to the charge-storing materials. This should save time and resources for researchers and product developers alike.”
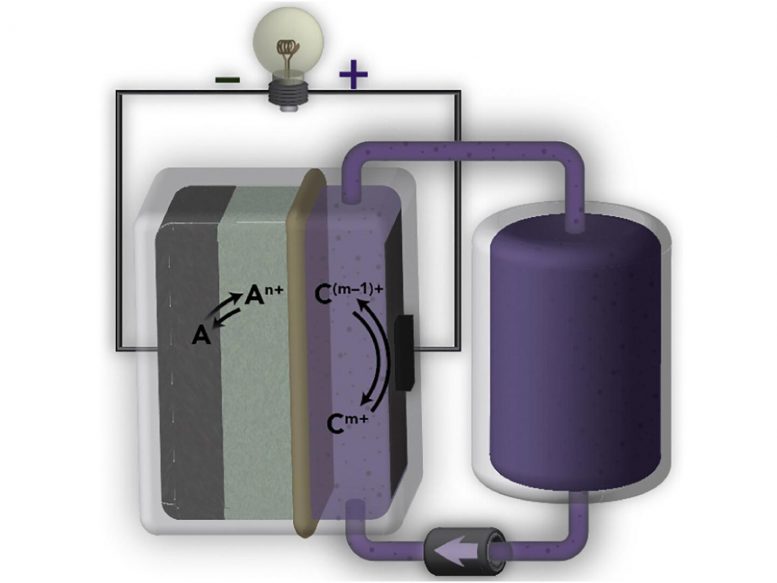
Most grid battery chemistries have highly alkaline (or basic) electrodes — a positively charged cathode on one side, and a negatively charged anode on the other side. But current state-of-the-art membranes are designed for acidic chemistries, such as the fluorinated membranes found in fuel cells, but not for alkaline flow batteries. (In chemistry, pH is a measure of the hydrogen ion concentration of a solution. Pure water has a pH of 7 and is considered neutral. Acidic solutions have a high concentration of hydrogen ions, and are described as having a low pH, or a pH below 7. On the other hand, alkaline solutions have low concentrations of hydrogen ions and therefore have a high pH, or a pH above 7. In alkaline batteries, the pH can be as high as 14 or 15.)
Fluorinated polymer membranes are also expensive. According to Helms, they can make up 15% to 20% of the battery’s cost, which can run in the range of $300/kWh.
One way to drive down the cost of flow batteries is to eliminate the fluorinated polymer membranes altogether and come up with a high-performing yet cheaper alternative such as AquaPIMs, said Miranda Baran, a graduate student researcher in Helms’ research group and the study’s lead author. Baran is also a Ph.D. student in the Department of Chemistry at UC Berkeley.
Getting back to basics
Helms and co-authors discovered the AquaPIM technology — which stands for “aqueous-compatible polymers of intrinsic microporosity” — while developing polymer membranes for aqueous alkaline (or basic) systems as part of a collaboration with co-author Yet-Ming Chiang, a principal investigator in JCESR and Kyocera Professor of Materials Science and Engineering at the Massachusetts Institute of Technology (MIT).
Through these early experiments, the researchers learned that membranes modified with an exotic chemical called an “amidoxime” allowed ions to quickly travel between the anode and cathode.

Later, while evaluating AquaPIM membrane performance and compatibility with different grid battery chemistries — for example, one experimental setup used zinc as the anode and an iron-based compound as the cathode — the researchers discovered that AquaPIM membranes lead to remarkably stable alkaline cells.
In addition, they found that the AquaPIM prototypes retained the integrity of the charge-storing materials in the cathode as well as in the anode. When the researchers characterized the membranes at Berkeley Lab’s Advanced Light Source (ALS), the researchers found that these characteristics were universal across AquaPIM variants.
Baran and her collaborators then tested how an AquaPIM membrane would perform with an aqueous alkaline electrolyte. In this experiment, they discovered that under alkaline conditions, polymer-bound amidoximes are stable — a surprising result considering that organic materials are not typically stable at high pH.
Such stability prevented the AquaPIM membrane pores from collapsing, thus allowing them to stay conductive without any loss in performance over time, whereas the pores of a commercial fluoropolymer membrane collapsed as expected, to the detriment of its ion transport properties, Helms explained.
This behavior was further corroborated with theoretical studies by Artem Baskin, a postdoctoral researcher working with David Prendergast, who is the acting director of Berkeley Lab’s Molecular Foundry and a principal investigator in JCESR along with Chiang and Helms.
Baskin simulated structures of AquaPIM membranes using computational resources at Berkeley Lab’s National Energy Research Scientific Computing Center (NERSC) and found that the structure of the polymers making up the membrane was significantly resistant to pore collapse under highly basic conditions in alkaline electrolytes.
A screen test for better batteries
While evaluating AquaPIM membrane performance and compatibility with different grid battery chemistries, the researchers developed a model that tied the performance of the battery to the performance of various membranes. This model could predict the lifetime and efficiency of a flow battery without having to build an entire device. They also showed that similar models could be applied to other battery chemistries and their membranes.
“Typically, you’d have to wait weeks if not months to figure out how long a battery will last after assembling the entire cell. By using a simple and quick membrane screen, you could cut that down to a few hours or days,” Helms said.
The researchers next plan to apply AquaPIM membranes across a broader scope of aqueous flow battery chemistries, from metals and inorganics to organics and polymers. They also anticipate that these membranes are compatible with other aqueous alkaline zinc batteries, including batteries that use either oxygen, manganese oxide, or metal-organic frameworks as the cathode.
Researchers from Berkeley Lab, UC Berkeley, Massachusetts Institute of Technology, and Istituto Italiano di Tecnologia participated in the study.
###
Reference: “Design Rules for Membranes from Polymers of Intrinsic Microporosity for Crossover-free Aqueous Electrochemical Devices” by Miranda J. Baran, Miles N. Braten, Swagat Sahu, Artem Baskin, Stephen M. Meckler, Longjun Li, Lorenzo Maserati, Mark E. Carrington, Yet-Ming Chiang, David Prendergast and Brett A. Helms, 10 October 2019, Joule.
DOI: 10.1016/j.joule.2019.08.025
This work was supported by the Joint Center for Energy Storage Research (JCESR), an Energy Innovation Hub funded by the U.S. Department of Energy, Office of Science. Additional funding was provided by the Center for Gas Separations Relevant to Clean Energy Technologies, a DOE Office of Science Energy Frontier Research Center.
Portions of the work, including polymer synthesis and characterization, were carried out at Berkeley Lab’s Molecular Foundry, a DOE Office of Science User Facility that specializes in nanoscale science.
The study also used GIWAXS (grazing-incidence wide-angle X-ray scattering) instruments at the ALS to characterize the AquaPIMs, and supercomputing resources at NERSC to model the polymer. The ALS and NERSC are DOE Office of Science User Facilities.
Never miss a breakthrough: Join the SciTechDaily newsletter.
Follow us on Google and Google News.
1 Comment
The problem with storage technology companies, is they only want to pursue grid scale solutions. PV was made cost effective by the purchases of the hardware by individuals, not utilities. Electrical storage should be made cost effective in the same way.
Utilities want to get rid of net metering. Utilities should be careful what they wish for. Net metering keeps them in the picture, as something beyond a transmission company. They might get what they wish for, and they might get it good and hard.