By combining two common polymers, polyacrylamide, and alginate, scientists at Harvard have created a new hydrogel that is capable of maintaining its elasticity and toughness over multiple stretches, possibly finding use in artificial cartilage, soft robotics, optics, artificial muscle, and as a protective covering for wounds.
A team of experts in mechanics, materials science, and tissue engineering at Harvard has created an extremely stretchy and tough gel that may suggest a new method for replacing damaged cartilage in human joints.
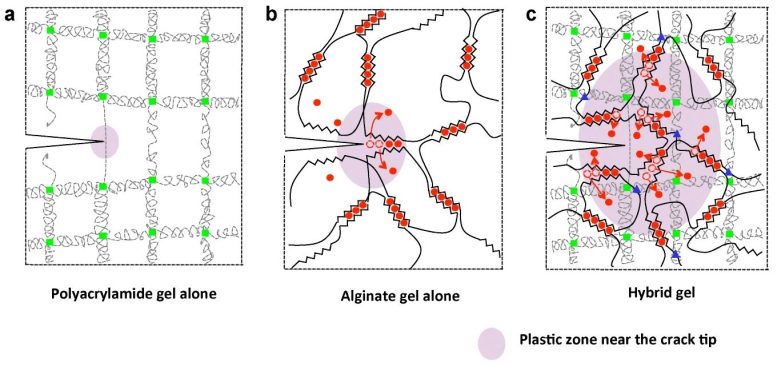
The new material, a hydrogel, is a strong hybrid of two weak gels. Not only can it stretch to 21 times its original length, but it is also tough, self-healing, and biocompatible — attributes that open up new opportunities in medicine and tissue engineering.
The material, its properties, and a simple method of synthesis are described in the September 6 issue of Nature.
“Conventional hydrogels are very weak and brittle — imagine a spoon breaking through jelly,” said lead author Jeong-Yun Sun, a postdoctoral fellow at the Harvard School of Engineering and Applied Sciences (SEAS). “But because [these gels] are water-based and biocompatible, people would like to use them for some very challenging applications like artificial cartilage or spinal disks. For a gel to work in those settings, it has to be able to stretch and expand under compression and tension without breaking.”

Sun and his co-authors were led by Professors Zhigang Suo, Joost J. Vlassak, and David J. Mooney.
To create the new hydrogel, the researchers combined two common polymers. The primary component is polyacrylamide, known for its use in soft contact lenses and as the electrophoresis gel that separates DNA fragments in biology labs; the second component is alginate, a seaweed extract that is commonly used to thicken food.
Separately, these gels are both quite weak — alginate, for instance, can stretch to only 1.2 times its length before it breaks. Combined in an 8-to-1 ratio, however, the two polymers form a complex network of cross-linked chains that reinforce one another. The chemical structure of this network allows the molecules to pull apart very slightly over a large area instead of permitting the gel to crack.
The alginate portion of the gel consists of polymer chains that form weak ionic bonds with one another, capturing calcium ions (added to the water) in the process. When the gel is stretched, some of the bonds between chains break — or “unzip,” as the researchers put it — releasing the calcium. As a result, the gel expands slightly, but the polymer chains themselves remain intact. Meanwhile, the polyacrylamide chains form a gridlike structure that bonds tightly with the alginate chains.
If the gel acquires a tiny crack as it stretches, the polyacrylamide grid helps to spread the pulling force over a large area, tugging on the alginate’s ionic bonds and unzipping them here and there. The research team showed that even with a huge crack, the hybrid gel can still stretch to 17 times its initial length.

Importantly, the new hydrogel is capable of maintaining its elasticity and toughness over multiple stretches. Provided the gel has time to relax between stretches, the ionic bonds between the alginate and the calcium can “re-zip.” The researchers have shown that raising the ambient temperature can accelerate this process.
The researchers’ combined expertise in mechanics, materials science, and bioengineering enabled them to apply two concepts from mechanics — crack bridging and energy dissipation — to a new problem.
“The unusually high stretchability and toughness of this gel, along with recovery, are exciting,” said Suo. “Now that we’ve demonstrated that this is possible, we can use it as a model system for studying the mechanics of hydrogels further, and explore various applications.”
Beyond artificial cartilage, the researchers see potential for the new hydrogel in soft robotics, in optics, in artificial muscle, and as a protective covering for wounds.
Reference: “Highly stretchable and tough hydrogels” by Jeong-Yun Sun, Xuanhe Zhao, Widusha R. K. Illeperuma, Ovijit Chaudhuri, Kyu Hwan Oh, David J. Mooney, Joost J. Vlassak and Zhigang Suo, 5 September 2012, Nature.
DOI: 10.1038/nature11409
Never miss a breakthrough: Join the SciTechDaily newsletter.
Follow us on Google and Google News.
1 Comment
Fascinating.