Nasal nanodrops carrying gold-based spherical nucleic acids can slip into the brain and activate powerful immune pathways that target glioblastoma.
In mice, the treatment cleared tumors and produced long-lasting protection when used with T-cell-boosting drugs.
Noninvasive Nanomedicine Breakthrough for Deadly Brain Cancer
Researchers at Washington University School of Medicine in St. Louis, working with partners at Northwestern University, have created a noninvasive strategy aimed at treating one of the most dangerous and fast-moving brain cancers. Their approach relies on carefully engineered nanoscale structures that carry powerful anti-tumor compounds directly into the brain through simple nasal drops. In tests with mice, this delivery method successfully targeted glioblastoma and strengthened the brain’s immune defenses, while remaining less invasive than many similar therapies now being explored.
The findings were published this month in PNAS.
Understanding Glioblastoma’s Rapid Progression and Treatment Barriers
Glioblastoma begins in astrocytes, a type of brain cell, and is the most common malignant brain tumor in the United States, affecting about three in every 100,000 people. It grows rapidly and is almost always fatal. One major barrier to treating it is the difficulty of safely transporting therapeutic drugs into the brain.
“We wanted to change this reality and develop a noninvasive treatment that activates the immune response to attack glioblastoma,” said Alexander H. Stegh, PhD, a professor and vice chair of research in the WashU Medicine Taylor Family Department of Neurosurgery and co-corresponding author of the study. Stegh also is research director of The Brain Tumor Center at Siteman Cancer Center, based at Barnes-Jewish Hospital and WashU Medicine. “With this research, we’ve shown that precisely engineered nanostructures, called spherical nucleic acids, can safely and effectively activate powerful immune pathways within the brain. This redefines how cancer immunotherapy can be achieved in otherwise difficult-to-access tumors.”
Turning “Cold Tumors” Hot With STING Pathway Activation
Glioblastoma tumors are sometimes described as “cold tumors” because they do not naturally trigger a strong immune reaction. This makes them more difficult to treat than “hot tumors,” which respond better to immunotherapies. To overcome this, scientists have been exploring ways to activate a cellular pathway called STING, short for stimulator of interferon genes. When a cell detects foreign DNA, STING helps launch an immune response.
Earlier research found that drugs designed to activate STING can help prepare the immune system to recognize and attack glioblastoma. However, these drugs deteriorate quickly in the body and only work effectively when delivered directly into the tumor. Achieving the repeated dosing needed for sustained benefit requires highly invasive procedures.
“We really wanted to minimize patients having to go through that when they are already ill, and I thought that we could use the spherical nucleic acid platforms to deliver these drugs in a noninvasive way,” said Akanksha Mahajan, PhD, a postdoctoral research associate in Stegh’s lab and the first author on the study.
Nasal-to-Brain Delivery Proves Effective in Mouse Models
To overcome the problem, the Stegh team collaborated with co-corresponding author Chad A. Mirkin, PhD, director of the International Institute for Nanotechnology and the Rathmann Professor of Chemistry at Northwestern University, and his team. Mirkin invented spherical nucleic acids, a class of nanostructures that arrange DNA or RNA densely around a nanoparticle core, and he has shown that they have greater therapeutic potency compared to the standard delivery methods. The WashU Medicine and Northwestern researchers prepared a new class of spherical nucleic acids with gold cores studded with short snippets of DNA to trigger activation of the STING pathway in specific immune cells. To deliver these drugs to the brain, the team turned to the nose.
Intranasal therapy has been explored as a potential delivery method for medications targeting the brain, but no nanoscale therapies had yet been developed using this method to activate immune responses against brain cancers.
STING Activation Spurs Potent Antitumor Immunity
“This is the first time that it has been shown that we can increase immune cell activation in glioblastoma tumors when we deliver nanoscale therapeutics from the nose to the brain,” Mahajan said.
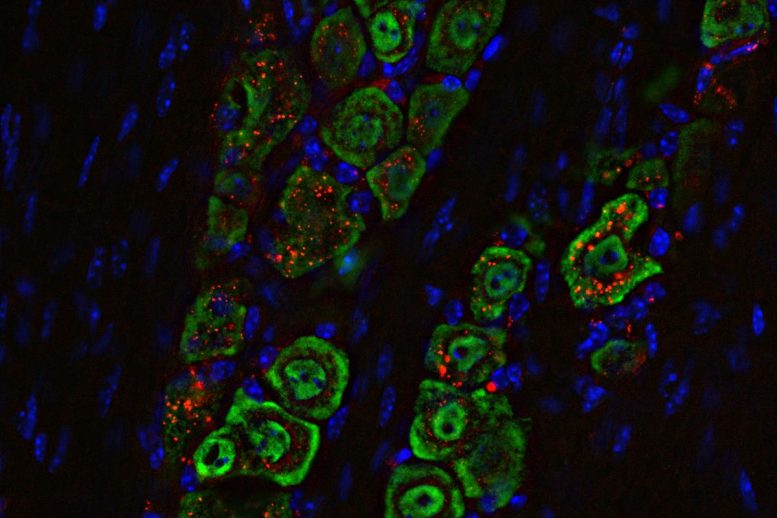
The team wanted to show that this approach could be used to deliver the medicine selectively to the brain, and that it would act on the appropriate cells once it got there. For the first objective, they used a molecular tag on the spherical nucleic acid that was visible under near-infrared light. They found that the nanomedicine, when delivered as droplets into the nasal passages of mice with glioblastoma, traveled along the path of the main nerve that connects facial muscles to the brain. The immune response evoked in the brain by the medicine was concentrated in the specific immune cells, especially those in the tumor itself, and triggered some helpful responses in the lymph nodes. The medicine did not spread to other parts of the body where it might cause unwanted side effects.
Examinations of immune cells in and near the tumor showed that the therapy successfully activated the STING pathway and armed the immune system to fight the tumor.
Clinical Promise and Next Steps Toward Translation
When applied in combination with drugs designed to help activate T lymphocytes, another type of immune cell, the new therapy eradicated the tumors with just one or two doses and induced long-term immunity against their recurrence. Taken together, the results were much better than those of current STING-activating immune therapies.
Stegh cautioned that firing up the STING pathway isn’t capable of curing glioblastomas without reinforcement from other therapeutic approaches. Turning on the STING pathway by itself isn’t enough to fight glioblastoma, because the tumor has many ways to block or shut down the immune response that STING is meant to activate. His team is looking to add capabilities to their nanostructure that activate other immune responses. This could allow physicians to double or triple the therapeutic targets all in a single therapy.
“This is an approach that offers hope for safer, more effective treatments for glioblastoma and potentially other immune treatment-resistant cancers, and it marks a critical step toward clinical application,” said Stegh.
Reference: “cGAS-agonistic spherical nucleic acids reprogram the glioblastoma immune microenvironment and promote antitumor immunity” by Akanksha S. Mahajan, Corey Dussold, Seunghyun Kim, Rachel Jarvis, Lisa A. Hurley, Serena Tommasini-Ghelfi, Jungsoo Park, Connor M. Forsyth, Bin Zhang, Jason Miska, Amy B. Heimberger, Chad A. Mirkin and Alexander H. Stegh, 3 November 2025, Proceedings of the National Academy of Sciences.
DOI: 10.1073/pnas.2409557122
This work was supported by the National Cancer Institute of the NIH (grant numbers P50CA221747 and R01CA275430), the NIH (grants R01CA120813, R01NS120547, and R01CA272639), the Melanoma Research Foundation, the Chicago Cancer Baseball Charities at the Lurie Cancer Center of Northwestern University and grants from Cellularity, Alnylam, and AbbVie. Imaging at Siteman Cancer Center Small Animal Cancer Imaging was supported by NIH instrumentation grants S10OD027042, S10OD025264, and National Cancer Institute Cancer Center grant P30CA091842. PET and MRI imaging was supported by Robert H. Lurie Comprehensive Cancer Center Grant P30CA060553.
Competing interests: Alexander Stegh is a shareholder of Exicure Inc., which develops SNA therapeutic platforms. Mirkin is a shareholder in Flashpoint, which develops SNA-based therapeutics. Stegh and Mirkin are co-inventors on patent US20150031745A1, which describes SNA nanoconjugates to cross the blood-brain barrier.
Never miss a breakthrough: Join the SciTechDaily newsletter.
Follow us on Google and Google News.