Federal funding awarded for rapid COVID-19 test based on a new biolabeling technology called plasmonic-fluor.
Engineers at the McKelvey School of Engineering at Washington University in St. Louis have received federal funding for a rapid COVID-19 test using a newly developed technology.
Srikanth Singamaneni, professor of mechanical engineering and materials science, and his team have developed a rapid, highly sensitive, and accurate biosensor based on an ultrabright fluorescent nanoprobe, which has the potential to be broadly deployed.
Called plasmonic-fluor, the ultrabright fluorescent nanoprobe can also help in resource-limited conditions because it requires fewer complex instruments to read the results. The National Science Foundation has awarded Singamaneni and his team a $100,008 grant toward developing a COVID-19 test using plasmonic-fluor.
Singamaneni hypothesizes their plasmonic-fluor-based biosensor will be 100 times more sensitive compared with the conventional SARS-CoV-2 antibody detection method. Increased sensitivity would allow clinicians and researchers to more easily find positive cases and lessen the chance of false negatives.
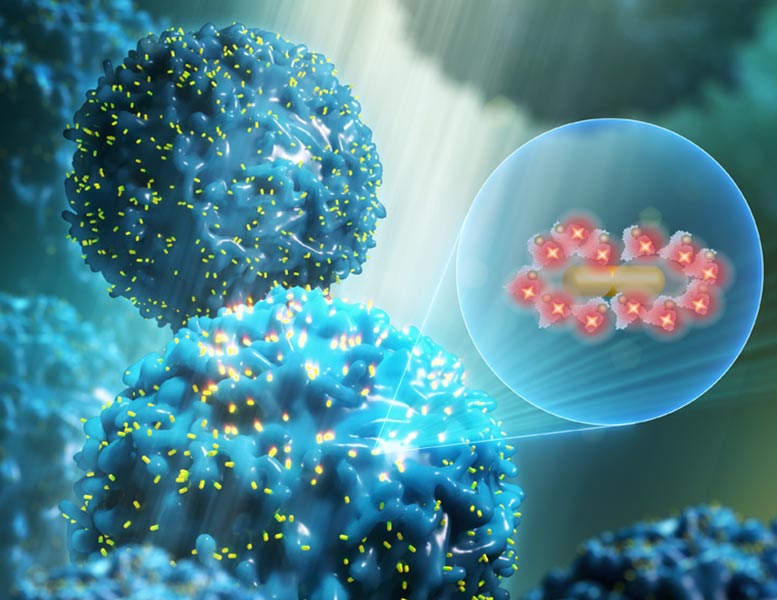
Plasmonic-fluor works by increasing the fluorescence signal to background noise. Imagine trying to catch fireflies outside on a sunny day. You might net one or two, but against the glare of the sun, those little buggers are difficult to see. What if those fireflies had the similar brightness as a high-powered flashlight?
Plasmonic-fluor effectively turns up the brightness of fluorescent labels used in a variety of biosensing and bioimaging methods. In addition to COVID-19 testing, it could potentially be used to diagnose, for instance, that a person has had a heart attack by measuring the levels of relevant molecules in blood or urine samples.
Using plasmonic-fluor, which is composed of gold nanoparticles coated with conventional dyes, researchers have been able to achieve up to a 6,700-fold brighter fluorescent nanolabel compared with conventional dyes, which can potentially lead to early diagnosis. Using this nanolabel as an ultrabright flashlight, they have demonstrated the detection of extremely small amounts of target biomolecules in biofluids and even molecules present on the cells.
The study was published in the April 20, 2020, issue of Nature Biomedical Engineering.
Gold nanoparticles serve as beacons
In biomedical research and clinical labs, fluorescence is used as a beacon to see and follow target biomolecules with precision. It’s an extremely useful tool, but it’s not perfect.
“The problem in fluorescence is, in a lot of cases, it’s not sufficiently intense,” Singamaneni said. If the fluorescent signal isn’t strong enough to stand out against background signals, just like fireflies against the glare of the sun, researchers may miss seeing something less abundant but important.
“Increasing the brightness of a nanolabel is extremely challenging,” said Jingyi Luan, lead author of the paper. But here, it’s the gold nanoparticle sitting at the center of the plasmonic-fluor that really does the work of efficiently turning the fireflies into flashlights, so to speak. The gold nanoparticle acts as an antenna, strongly absorbing and scattering light. That highly concentrated light is funneled into the fluorophore placed around the nanoparticle. In addition to concentering the light, the nanoparticles speed up the emission rate of the fluorophores. Taken together, these two effects increase the fluorescence emission.
Essentially, each fluorophore becomes a more efficient beacon, and the 200 fluorophores sitting around the nanoparticle emit a signal that is equal to 6,700 fluorophores.
In addition to detecting low quantities of molecules, sensing time can be shortened using plasmonic-fluor as brighter beacons mean fewer captured proteins are needed to determine their presence.
The researchers have also shown that plasmonic-fluor allows the detection of multiple proteins simultaneously. And in flow cytometry, plasmonic-fluor’s brightening effect allows for a more precise and sensitive measurement of proteins on cell surface, whose signal may have been buried in the background noise using traditional fluorescent tagging.
There have been other efforts to enhance fluorescent tagging in imaging, but many require the use of an entirely new workflow and measurement platform. In addition to plasmonic-fluor’s ability to greatly increase the sensitivity and decrease the sensing time, it doesn’t require any changes to existing laboratory tools or techniques.
Reference: “Ultrabright fluorescent nanoscale labels for the femtomolar detection of analytes with standard bioassays” by Jingyi Luan, Anushree Seth, Rohit Gupta, Zheyu Wang, Priya Rathi, Sisi Cao, Hamed Gholami Derami, Rui Tang, Baogang Xu, Samuel Achilefu, Jeremiah J. Morrissey and Srikanth Singamaneni, 20 April 2020, Nature Biomedical Engineering.
DOI: 10.1038/s41551-020-0547-4
The technology has been licensed to Auragent Bioscience LLC by Washington University’s Office of Technology Management. Auragent is in the process of further development and scaling up the production of plasmonic-fluors for commercialization.
Never miss a breakthrough: Join the SciTechDaily newsletter.
Follow us on Google and Google News.