A microscopic flaw in the brain’s cellular scaffolding can shape brain size for life.
Why do some children develop a brain that is smaller than expected (microcephaly)? An international team of scientists from the German Primate Center – Leibniz Institute for Primate Research (DPZ), Hannover Medical School (MHH), and the Max Planck Institute of Molecular Cell Biology and Genetics investigated this question using human brain organoids. These lab-grown models allowed the researchers to study how changes in essential structural proteins inside cells can disrupt early brain development and lead to this serious condition (EMBO Reports).
The researchers found that mutations in actin genes interfere with how early brain progenitor cells divide. When these cells do not divide properly, fewer of them are produced. This shortage limits overall brain growth and results in a smaller brain. “Our findings provide the first cellular explanation for microcephaly in people with the rare Baraitser-Winter syndrome,” says Indra Niehaus, first author of the study and research associate at Hannover Medical School.
How the Cell’s Internal Support System Affects Brain Development
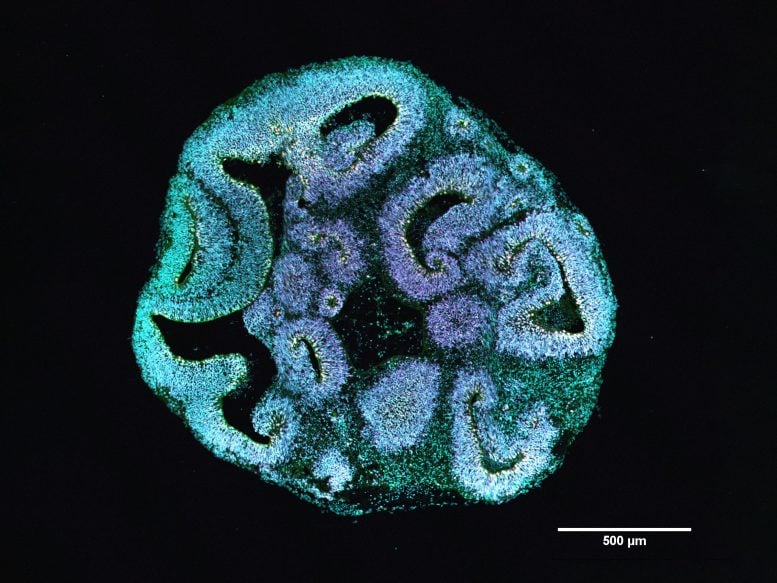
Actin is a core component of the cytoskeleton, which serves as the internal framework that gives cells structure and helps move materials within them. People with Baraitser-Winter syndrome carry a mutation in one of two key actin genes. To study the effects of these mutations, the scientists reprogrammed skin cells from affected patients into induced pluripotent stem cells. These cells were then used to grow three-dimensional brain organoids that reproduce critical stages of early human brain development.
The differences between patient-derived organoids and those from healthy donors were evident after thirty days. Organoids carrying the actin mutation were roughly 25 percent smaller. The ventricle-like regions inside the organoids, which house progenitor cells and give rise to early nerve cells, were also much smaller.
A Decline in Key Brain Building Cells
When the team analyzed the types of cells inside the organoids, they observed a clear shift in cell populations. Apical progenitor cells, which play a central role in forming the cerebral cortex, were present in much lower numbers. At the same time, basal progenitor cells increased. These cells are normally produced later in development, suggesting that the normal developmental timeline had been disrupted.
This imbalance pointed to problems in how progenitor cells were dividing and renewing themselves.
When Cell Division Orientation Changes
Using high-resolution microscopy, the researchers closely examined how apical progenitor cells divided. Under normal conditions, these cells divide mainly at right angles to the ventricular zone. This orientation allows cellular components to be evenly distributed and produces two new apical progenitor cells.
In organoids with actin mutations, this process was altered. Vertical divisions became far less common, while horizontal and angled divisions occurred more frequently. Because of this shift, apical progenitor cells were less likely to renew themselves. Many detached from the ventricular zone and instead became basal progenitor cells.
“Our analyses show very clearly that a change in the division orientation of the progenitor cells is the decisive trigger for the reduced brain size,” says Michael Heide, group leader at the German Primate Center and last author of the study. “A single change in the cytoskeleton is sufficient to disrupt the course of early brain development.”
Small Structural Changes With Big Effects
Electron microscopy revealed additional abnormalities at the ventricular surface. Cell shapes were uneven, and extra protrusions appeared between neighboring cells. The researchers also found unusually high levels of tubulin at cell junctions. Tubulin is another cytoskeletal protein that plays an important role in cell division.
Although the overall structure of the cells remained intact, these subtle changes may be enough to permanently interfere with how cells orient themselves during division.
Confirming the Genetic Cause
To make sure the observed differences were not caused by unrelated genetic factors, the team performed a key control experiment. Using CRISPR/Cas9, they introduced the same actin mutation found in Baraitser-Winter syndrome patients into a healthy stem cell line. Brain organoids grown from these edited cells developed the same defects seen in patient-derived organoids, confirming that the mutation itself is responsible.
What the Findings Mean for Medicine
The results offer new insight into how rare genetic mutations can lead to complex brain malformations and demonstrate the power of brain organoids in biomedical research. “Our findings help us understand how rare genetic disorders lead to complex brain malformations and highlight the potential of brain organoids for biomedical research,” says Michael Heide.
“The therapeutic potential of this study lies in diagnostics, as our data helps to better classify genetic findings in patients. Since the disease affects early fetal development processes, interventions in humans would be complex. However, new drugs that influence the interaction between actin and microtubules could open up new approaches in the long term,” says Nataliya Di Donato, Director of the Institute of Human Genetics at Hannover Medical School.
Reference: “Cerebral organoids expressing mutant actin genes reveal cellular mechanism underlying microcephaly” by Indra Niehaus, Michaela Wilsch-Bräuninger, Felipe Mora-Bermúdez, Fabian Rost, Mihaela Bobic-Rasonja, Velena Radosevic, Marija Milkovic-Perisa, Pauline Wimberger, Mariasavina Severino, Alexandra Haase, Ulrich Martin, Karolina Kuenzel, Kaomei Guan, Katrin Neumann, Noreen Walker, Evelin Schröck, Natasa Jovanov-Milosevic, Wieland B Huttner, Nataliya Di Donato and Michael Heide, 10 December 2025, EMBO Reports.
DOI: 10.1038/s44319-025-00647-7
Never miss a breakthrough: Join the SciTechDaily newsletter.
Follow us on Google and Google News.
1 Comment
Autism . Help ….. proteins…… thank you