Defective mitochondria – the ‘batteries’ that power the cells of our bodies – could in the future be repaired using gene-editing techniques. Scientists at the University of Cambridge have shown that it is possible to modify the mitochondrial genome in live mice, paving the way for new treatments for incurable mitochondrial disorders.
Our cells contain mitochondria, which provide the energy for our cells to function. Each of these mitochondria contains a tiny amount of mitochondrial DNA. Mitochondrial DNA makes up only 0.1% of the overall human genome and is passed down exclusively from mother to child.
Faults in our mitochondrial DNA can affect how well the mitochondria operate, leading to mitochondrial diseases, serious and often fatal conditions that affect around 1 in 5,000 people. The diseases are incurable and largely untreatable.
“[This] shows that, in principle, we can go in and correct spelling mistakes in defective mitochondrial DNA, producing healthy mitochondria that allow the cells to function properly.” — Pedro Silva-Pinheiro
There are typically around 1,000 copies of mitochondrial DNA in each cell, and the percentage of these that are damaged, or mutated, will determine whether a person will suffer from mitochondrial disease or not. Usually, more than 60% of the mitochondria in a cell need to be faulty for the disease to emerge, and the more defective mitochondria a person has, the more severe their disease will be. If the percentage of defective DNA could be reduced, the disease could potentially be treated.
A cell that contains a mixture of healthy and faulty mitochondrial DNA is described as ‘heteroplasmic’. If a cell contains no healthy mitochondrial DNA, it is ‘homoplasmic’.
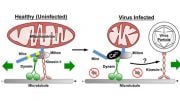
In 2018, a team from the MRC Mitochondrial Biology Unit at the University of Cambridge applied an experimental gene therapy treatment in mice and were able to successfully target and eliminate the damaged mitochondrial DNA in heteroplasmic cells, allowing mitochondria with healthy DNA to take their place.
“Our earlier approach is very promising and was the first time that anyone had been able to alter mitochondrial DNA in a live animal,” explained Dr. Michal Minczuk. “But it would only work in cells with enough healthy mitochondrial DNA to copy themselves and replace the faulty ones that had been removed. It would not work in cells whose entire mitochondria had faulty DNA.”
In their latest advance, published recently in Nature Communications, Dr. Minczuk and colleagues used a biological tool known as a mitochondrial base editor to edit the mitochondrial DNA of live mice. The treatment is delivered into the bloodstream of the mouse using a modified virus, which is then taken up by its cells. The tool looks for a unique sequence of base pairs – combinations of the A, C, G and T molecules that make up DNA. It then changes the DNA base – in this case, changing a C to a T. This would, in principle, enable the tool to correct certain ‘spelling mistakes’ that cause the mitochondria to malfunction.
There are currently no suitable mouse models of mitochondrial DNA diseases, so the researchers used healthy mice to test the mitochondrial base editors. However, it shows that it is possible to edit mitochondrial DNA genes in a live animal.
Pedro Silva-Pinheiro, a postdoctoral researcher in Dr. Minczuk’s lab and first author of the study, said: “This is the first time that anyone has been able to change DNA base pairs in mitochondria in a live animal. It shows that, in principle, we can go in and correct spelling mistakes in defective mitochondrial DNA, producing healthy mitochondria that allow the cells to function properly.”
An approach pioneered in the UK known as mitochondrial replacement therapy – sometimes referred to as ‘three-person IVF’ – allows a mother’s defective mitochondria to be replaced with those from a healthy donor. However, this technique is complex, and even standard IVF is successful in fewer than one in three cycles.
Dr. Minczuk added: “There’s clearly a long way to go before our work could lead to a treatment for mitochondrial diseases. But it shows that there is the potential for a future treatment that removes the complexity of mitochondrial replacement therapy and would allow for defective mitochondria to be repaired in children and adults.”
Reference: “In vivo mitochondrial base editing via adeno-associated viral delivery to mouse post-mitotic tissue” by Pedro Silva-Pinheiro, Pavel A. Nash, Lindsey Van Haute, Christian D. Mutti, Keira Turner and Michal Minczuk, 8 February 2022, Nature Communications.
DOI: 10.1038/s41467-022-28358-w
The research was funded by the Medical Research Council UK, the Champ Foundation and the Lily Foundation.




Be the first to comment on "Potential for Gene-Editing To Enable New Treatments for Incurable Mitochondrial Disorders"