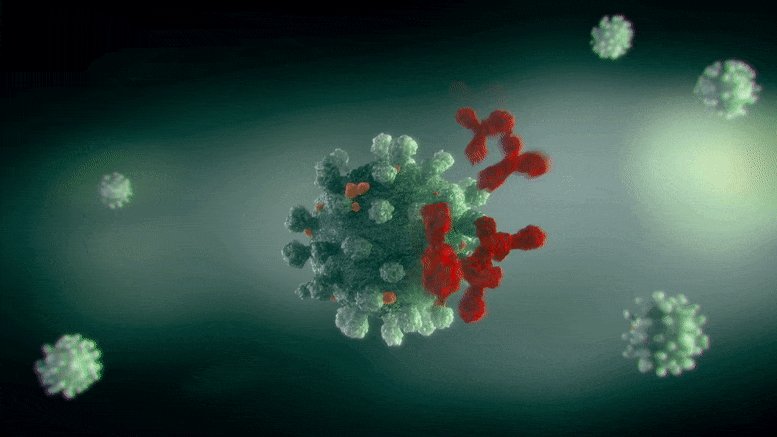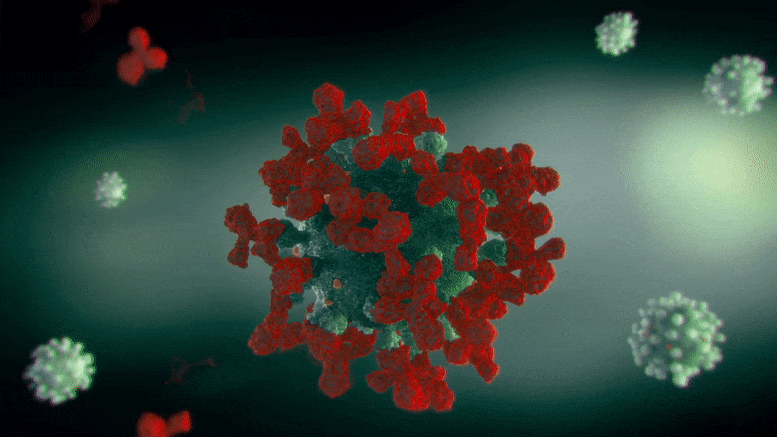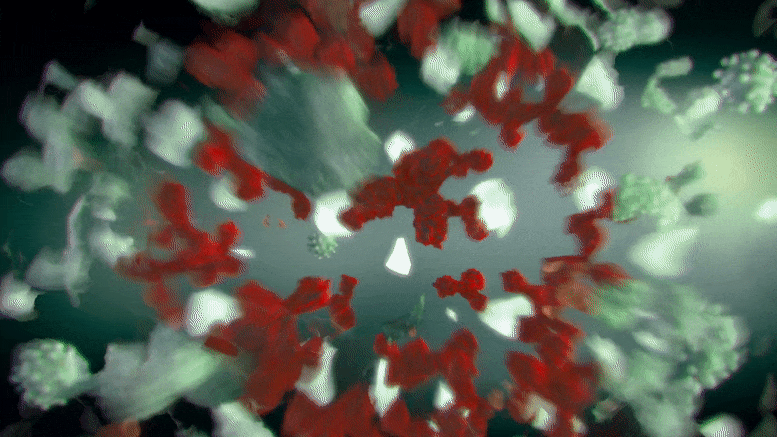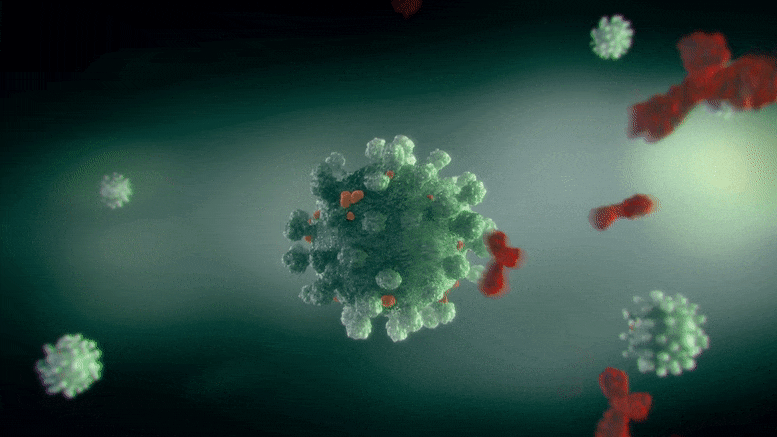
The Scripps Research team demonstrated that a variant of an existing FDA-approved treatment for neurological disorders may prevent COVID-19 infection in animals.
Scientists at Scripps Research Institute have created a new drug that may make the COVID-19 virus into a harbinger of its own doom.
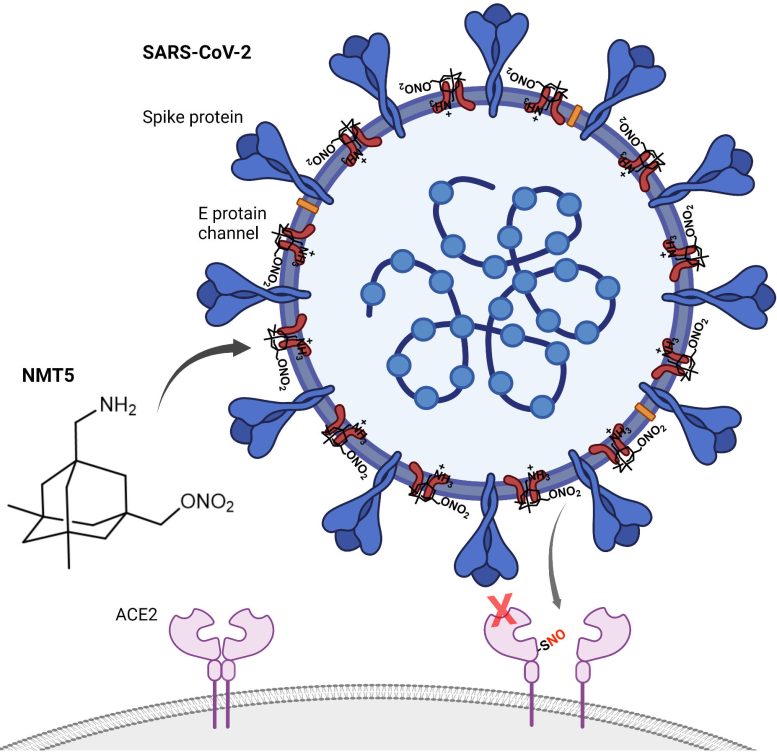
The drug, NMT5, described in a recent paper published in Nature Chemical Biology, coats SARS-CoV-2 with chemicals that may temporarily modify the human ACE2 receptor—the molecule the virus normally latches onto to infect cells. That means that while the virus is nearby, its entry into human cells through the ACE2 receptor is inhibited; however, when the virus is not there, ACE2 may operate normally.
“What’s so neat about this drug is that we’re actually turning the virus against itself,” says senior author Stuart Lipton, MD, Ph.D., the Step Family Endowed Chair and Scripps Research professor. “We’re arming it with little molecular warheads that end up preventing it from infecting our cells; it’s our revenge on the virus.”
Blocking ACE2 When Virus Is Near
Lipton and his colleagues have long been researching variations of the drug memantine, which Lipton created and patented in the 1990s for treating neurological diseases such as Alzheimer’s. While memantine originated from an anti-influenza drug in the 1960s, clinicians began looking into it for other disorders after seeing a woman with Parkinson’s symptoms improve when she took the drug for the flu.
“My team had made these antiviral drugs better for the brain, and when COVID-19 emerged, we wondered whether we had also, in the process, made any of them better antivirals,” says Lipton.
Lipton and his colleagues tested a library of compounds with overall structures comparable to memantine but with additional pharmacological warheads. They identified NMT5 as a drug candidate with two critical properties: it could detect and attach to a pore on the surface of SARS-CoV-2, and it could chemically modify human ACE2 using a nitroglycerin fragment as the warhead. The team realized that this could transform the virus into a vehicle for its own destruction.
In the new paper, Lipton’s group characterized and tested NMT5 in isolated cells as well as animals. They showed how NMT5 attaches tightly to SARS-CoV-2 viral particles as the viruses move through the body. Then, they revealed the details of how the drug adds a chemical (similar to nitroglycerin) to certain molecules if it gets close enough. When the virus gets near ACE2 to infect a cell, that translates into NMT5 adding a “nitro group” to the receptor. When ACE2 is modified in this way, its structure temporarily shifts – for about 12 hours – so that the SARS-CoV-2 virus can no longer bind to it to cause infection.
“What’s really beautiful is that this only knocks down the availability of ACE2 locally when the virus is coming at it,” says Lipton. “It doesn’t knock down all the function of ACE2 elsewhere in the body, allowing for the normal function of this protein.”
Inhibiting Viral Binding and Reducing Inflammation
In cell culture experiments testing how well the Omicron variant of SARS-CoV-2 can attach to human ACE2 receptors, the drug prevented 95% of viral binding. In hamsters with COVID-19, NMT5 decreased virus levels by 100-fold, eliminated blood vessel damage in the animals’ lungs, and ameliorated inflammation. The drug also showed effectiveness against nearly a dozen other variants of COVID-19, including alpha, beta, gamma, and delta strains.
Most anti-viral drugs work by directly blocking part of a virus—which can pressure the virus to evolve resistance to the drug. Since NMT5 is only using the virus as a carrier, the researchers think the drug is likely to be effective against many other variants of SARS-CoV-2.
“We expect this compound would continue to be effective even as new variants emerge because it doesn’t rely on attacking parts of the virus that commonly mutate,” says Chang-ki Oh, a senior staff scientist and first author of the new paper.
Though they have only studied the compound in animal models, the team is now making a version of the drug to evaluate for human use, while carrying out additional safety and effectiveness trials in animals.
“These exciting findings suggest a new avenue for drug development that requires drug combinations for effective pandemic preparedness,” says co-author Arnab Chatterjee, Ph.D.
Reference: “Targeted protein S-nitrosylation of ACE2 inhibits SARS-CoV-2 infection” by Chang-ki Oh, Tomohiro Nakamura, Nathan Beutler, Xu Zhang, Juan Piña-Crespo, Maria Talantova, Swagata Ghatak, Dorit Trudler, Lauren N. Carnevale, Scott R. McKercher, Malina A. Bakowski, Jolene K. Diedrich, Amanda J. Roberts, Ashley K. Woods, Victor Chi, Anil K. Gupta, Mia A. Rosenfeld, Fiona L. Kearns, Lorenzo Casalino, Namir Shaabani, Hejun Liu, Ian A. Wilson, Rommie E. Amaro, Dennis R. Burton, John R. Yates III, Cyrus Becker, Thomas F. Rogers, Arnab K. Chatterjee and Stuart A. Lipton, 29 September 2022, Nature Chemical Biology.
DOI: 10.1038/s41589-022-01149-6
The study was funded by the Scripps Center Grant for Antiviral Medicines & Pandemic Preparedness (CAMPP AViDD) from the National Institutes of Health (U19 AI171443).
Never miss a breakthrough: Join the SciTechDaily newsletter.
Follow us on Google and Google News.
1 Comment
A nitro defense?
Hey… Am I trippin ‘on the last VAC or did Smith’s head just explode!?