A computational method accurately predicts the optimal ligand for a photochemical palladium catalyst, enabling new radical reactions of alkyl ketones.
Ketones are common components in many organic molecules, and chemists are continually exploring new ways to use them in forming chemical bonds. One particularly difficult reaction involves the one-electron reduction of ketones to produce ketyl radicals. These radicals are highly reactive intermediates that play a key role in natural product synthesis and pharmaceutical development.
However, most existing techniques work best for aryl ketones, while simple alkyl ketones remain difficult to manipulate. Although alkyl ketones are much more prevalent, their chemical structure makes them significantly harder to reduce.
In response to this challenge, a team of organic and computational chemists from WPI-ICReDD at Hokkaido University has created a new catalytic method that successfully generates alkyl ketyl radicals. Their findings were published on October 20, 2025, in the Journal of the American Chemical Society and are freely available as open-access research.
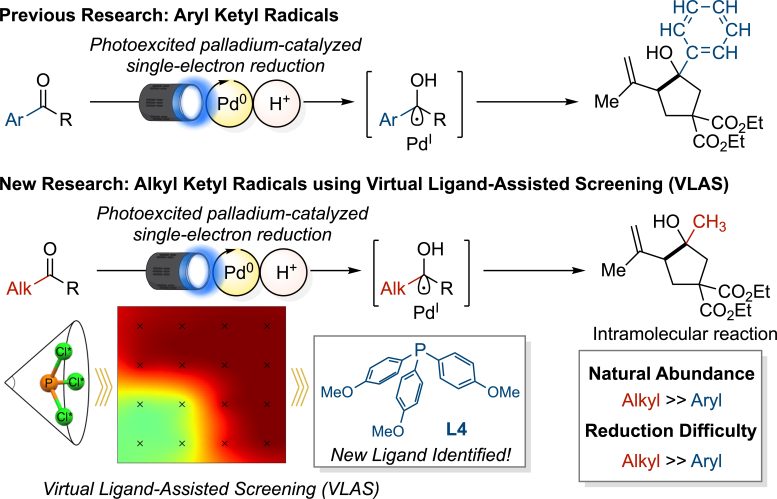
The WPI-ICReDD group had previously shown that a palladium catalyst paired with phosphine ligands could drive photochemical reactions (activated by shining light) with aryl ketones. However, this system did not work with alkyl ketones. Their experiments indicated that while alkyl ketyl radicals could initially form, they quickly transferred an electron back to the palladium catalyst—a process known as back electron transfer (BET)—before any further reaction could take place. As a result, the starting material remained unchanged.
Computational Chemistry to the Rescue
Just like in conventional palladium catalysis, the reactivity of photoexcited palladium catalysts depends greatly on the type of phosphine ligand used. Therefore, the team hypothesized they could identify an appropriate phosphine ligand capable of engendering reactivity towards alkyl ketones.
However, since thousands of phosphine ligands are known, identifying the optimal one for an unknown reaction through experimentation alone would be difficult, time-consuming, and environmentally burdensome due to chemical waste.

The researchers effectively circumvented these issues by utilizing computational chemistry to efficiently search for optimal ligands with minimal experiments. Specifically, they employed the Virtual Ligand-Assisted Screening (VLAS) method developed by Associate Professor Wataru Matsuoka and Professor Satoshi Maeda from WPI-ICReDD. For 38 different phosphine ligands, the VLAS generated a heat map that predicted which ligands could best engender reactivity based on their electronics and sterics.
Based on this heat map, the team selected just three promising ligands for experimental testing and successfully identified L4 as the optimal ligand—tris(4-methoxyphenyl)phosphine (P(p-OMe-C₆H₄)₃). Using this ligand effectively suppressed BET, enabling the generation of ketyl radicals from alkyl ketones and achieving versatile reactions with high yield.
This work provides chemists with facile access to alkyl ketyl radical reactivity and highlights the effectiveness of VLAS to rapidly develop and optimize new chemical reactions.
Reference: “Virtual Ligand-Assisted Screening for the Generation of Ketyl Radicals from Alkyl Ketones via Photoexcited Palladium Catalysis” by Kosaku Tanaka, III, Ren Yamada, Suvankar Debbarma, Wataru Kanna, Hiroki Hayashi, Wataru Matsuoka, Satoshi Maeda and Tsuyoshi Mita, 20 October 2025, Journal of the American Chemical Society.
DOI: 10.1021/jacs.5c13115
Funding: Japan Science and Technology Agency, Japan Society for the Promotion of Science
Never miss a breakthrough: Join the SciTechDaily newsletter.
Follow us on Google and Google News.
1 Comment
the missing property miloranical and burn… its a pattern of inrawa when your grid shows pattern of conocu..