Scientists at the Yale School of Engineering and Applied Sciences have developed a passive-flow microfluidic device for imaging latent HIV activation dynamics in single T cells.
While studying latent HIV cells, researchers in the laboratory of Prof. Kathryn Miller-Jensen have developed a device that could make it a lot easier to track changes in cells over a period of time.
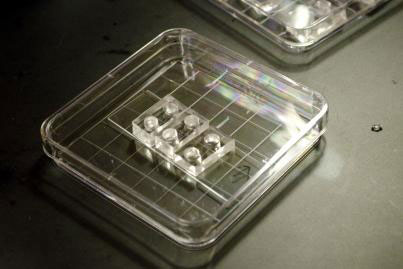
The device, which fits in the palm of a hand, is essentially two small reservoirs connected by a channel. Ramesh Ramji, a post doctoral researcher in Miller-Jensen’s lab, came up with the idea after a grad student had trouble studying some cells. They recently published a paper about the device in the journal Integrative Biology.
“He said this would work, and I thought, ‘I’m not sure – this seems too simple,'” said Miller-Jensen, who teaches Biomedical Engineering & Molecular, Cellular & Developmental Biology. “But it did.”
Her lab has filed a provisional patent on the device. Other labs at Yale are now using the device to image yeast, and one lab is using it to measure the nuclei isolate from nematode worms. She’s also heard from the representative of a company that screens HIV drugs seeking more information about the device.
Miller-Jensen’s lab is looking at T-cells, a kind of lymphocyte that grows in suspension – that is, they float freely in media. The trouble with suspension cells is that they’re always moving all around. You could adhere them to a plate, but this requires protein or chemicals on the surface, and could change their biology. That makes it difficult for the researchers to image the cells while they’re alive.
“So if you want to keep them in one place to stain them, and look at different things in the same cells repeatedly, it’s hard to do that,” Miller-Jensen said.
There are other devices that serve a similar purpose, but they require tubing, external pumps, and an elaborate process to operate.
“Here, you literally take a pipette and you just place the cells into the device,” she said. Due to the uneven heights of the fluid in each reservoir, the cells flow from one to the other (this is known as “passive flow,” caused by hydrostatic pressure, and it’s the reason pumps aren’t necessary). As they pass through the channel, they get caught in small traps – each one about five nanometers wide.
Although it has many potential uses, the device was specifically created in service to the work in Miller-Jensen’s lab on latent HIV cells. They want to learn more about why genetically identical cells with a latent HIV virus respond differently to drugs that are targeting them. The drugs activate some of the cells early in the process, others late in the process, and still others not at all.
Those variations have proven to be a key challenge to perfecting the “activate-and-kill” approach to latent HIV cells. The activate-and-kill theory posits that if you can get drugs to trigger all the latent cells, and then kill those cells, you can ultimately cure the patient. To get a better handle on why there are these variations in the cells, Miller-Jensen’s lab is tracking multiple events in the same cells to examine the biological events that happen over time after the application of drugs
“We’ve done that on a plate, but it’s difficult, and we could do more things if we could track the cells,” she said. “So this device is really pushing us forward in seeing if we could measure lots of the things in the same cell over time, and understand why one cell is different from another. And I think this would inform some of the molecular biology behind HIV anti-latency drug design.”
Reference: “A passive-flow microfluidic device for imaging latent HIV activation dynamics in single T cells” by Ramesh Ramji, Victor C. Wong, Arvind K. Chavali, Larisa M. Gearhartac and Kathryn Miller-Jensen, 26 June 2015, Integrative Biology.
DOI: 10.1039/C5IB00094G
Never miss a breakthrough: Join the SciTechDaily newsletter.
Follow us on Google and Google News.